1,5-i-aedans
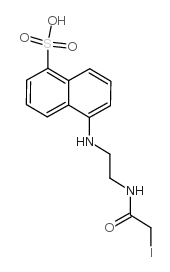
1,5-i-aedans structure
|
Common Name | 1,5-i-aedans | ||
|---|---|---|---|---|
| CAS Number | 36930-63-9 | Molecular Weight | 434.24900 | |
| Density | 1.808 g/cm3 | Boiling Point | N/A | |
| Molecular Formula | C14H15IN2O4S | Melting Point | 300ºC | |
| MSDS | Chinese USA | Flash Point | N/A | |
|
Detection of proteins on blot membranes.
Curr. Protoc. Protein Sci. Chapter 10 , Unit 10.8, (2001) Staining of blot transfer membranes permits visualization of proteins and allows the extent of transfer to be monitored. In the protocols described in this unit, proteins are stained after electroblotting from one-dimensional or two-dimensional polyacrylamide... |
|
|
Iron-sulfur cluster biosynthesis: characterization of IscU-IscS complex formation and a structural model for sulfide delivery to the [2Fe-2S] assembly site.
J. Biol. Inorg. Chem. 14(6) , 829-39, (2009) Recent work on the bacterial iron-sulfur cluster (isc) family of gene products, and eukaryotic homologs, has advanced the molecular understanding of cellular mechanisms of iron-sulfur cluster biosynthesis. Members of the IscS family are pyridoxyl-5'-phosophat... |
|
|
Characterization of the formation of amyloid protofibrils from barstar by mapping residue-specific fluorescence dynamics.
J. Mol. Biol. 358(4) , 935-42, (2006) The small protein barstar aggregates at low pH to form soluble oligomers, which can be transformed into fibrillar aggregates at an elevated temperature. To characterize structurally, with residue-specific resolution, the process of amyloid formation of barsta... |
|
|
Fluorescence-based peptide labeling and fractionation strategies for analysis of cysteine-containing peptides.
Anal. Chem. 77(14) , 4495-502, (2005) This study demonstrates that 1,5-I-AEDANS (5-({2-[(iodoacetyl)amino]ethyl}amino)naphthalene-1-sulfonic acid) can be used as a versatile fluorescence-based peptide quantification tool and provides readily interpretable tandem mass spectra for de novo peptide s... |
|
|
Decavanadate binding to a high affinity site near the myosin catalytic centre inhibits F-actin-stimulated myosin ATPase activity.
Biochemistry 43(18) , 5551-61, (2004) Decameric vanadate (V(10)) inhibits the actin-stimulated myosin ATPase activity, noncompetitively with actin or with ATP upon interaction with a high-affinity binding site (K(i) = 0.27 +/- 0.05 microM) in myosin subfragment-1 (S1). The binding of V(10) to S1 ... |
|
|
Characterization of low density lipoprotein receptor ligand interactions by fluorescence resonance energy transfer.
J. Lipid Res. 47(5) , 1091-6, (2006) The low density lipoprotein receptor (LDLR) is the prototype of a family of cell surface receptors involved in a wide range of biological processes. A soluble low density lipoprotein receptor (sLDLR) and a tryptophan (Trp)-deficient variant human apolipoprote... |
|
|
Analysis of protein adduction kinetics by quantitative mass spectrometry: competing adduction reactions of glutathione-S-transferase P1-1 with electrophiles.
Chem. Biol. Interact. 168(2) , 117-27, (2007) Defining the mechanisms and consequences of protein adduction is crucial to understanding the toxicity of reactive electrophiles. Application of tandem mass spectrometry and data analysis algorithms enables detection and mapping of chemical adducts at the lev... |
|
|
Phospholamban inhibits Ca-ATPase conformational changes involving the E2 intermediate.
Biochemistry 46(7) , 1999-2009, (2007) We have used steady-state fluorescence spectroscopy in combination with enzyme kinetic assays to test the hypothesis that phospholamban (PLB) stabilizes the Ca-ATPase in the E2 intermediate state. The cardiac muscle Ca-ATPase (SERCA2a) isoform was expressed e... |
|
|
Loop dynamics of the extracellular domain of human tissue factor and activation of factor VIIa.
Biophys. J. 96(2) , 681-92, (2009) In the crystal structure of the complex between the soluble extracellular domain of tissue factor (sTF) and active-site-inhibited VIIa, residues 91 and 92 in the Pro(79)-Pro(92) loop of sTF interact with the catalytic domain of VIIa. It is not known, however,... |
|
|
Fluorescence spectroscopic analysis of the proximity changes between the central helix of troponin C and the C-terminus of troponin T from chicken skeletal muscle.
Biochim. Biophys. Acta 1774(4) , 466-73, (2007) Recent structural studies of the troponin (Tn) core complex have shown that the regulatory head containing the N-lobe of TnC is connected to the IT arm by a flexible linker of TnC. The IT arm is a long coiled-coil formed by alpha-helices of TnI and TnT, plus ... |