雌二醇
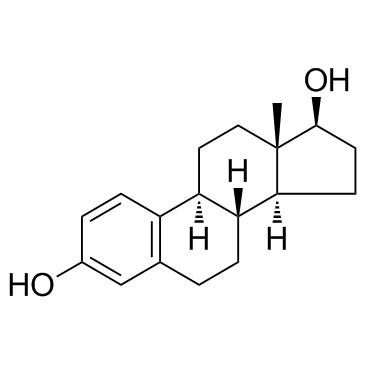
雌二醇结构式

|
常用名 | 雌二醇 | 英文名 | beta-Estradiol |
|---|---|---|---|---|
| CAS号 | 50-28-2 | 分子量 | 272.382 | |
| 密度 | 1.2±0.1 g/cm3 | 沸点 | 445.9±45.0 °C at 760 mmHg | |
| 分子式 | C18H24O2 | 熔点 | 173ºC | |
| MSDS | 中文版 美版 | 闪点 | 209.6±23.3 °C | |
| 符号 |

GHS08 |
信号词 | Danger |
雌二醇用途【用途一】 生化研究,雌性激素类药物。 【用途二】
雌激素类药。用于子宫功能性出血、原发性闭经、绝经期综合征、前列腺癌。能促进和调节女性性器官及副性征的正常发育。能促使乳腺导管发育增生,但是在剂量较大时,能抑制垂体前叶催乳素的释放,而减少乳汁分泌。也可用于回奶。但是可能有恶心、呕吐及子宫内膜过度增生而出血等。肝、肾功能不全者慎用。忌用于乳房、外阴和阴道粘膜。 【用途三】 雌二醇戊酸酯、雌二醇苯甲酸酯的中间体。 【用途四】 经前期卵巢分泌的只要雌激素。在青春期靠调控基因转录和蛋白质合成指导雌性表现型和胚胎的发育,也诱导促性腺激素的生成,诱导排卵。 更多
|
| 中文名 | 雌二醇 |
|---|---|
| 英文名 | 17β-estradiol |
| 中文别名 | 17β-雌二醇 | β-雌二醇 |
| 英文别名 | 更多 |
| 描述 | Estradiol 是一种类固醇性激素,对维持女性的生育能力和第二性征至关重要。 |
|---|---|
| 相关类别 | |
| 靶点 |
Human Endogenous Metabolite |
| 体外研究 | 雌二醇在海马CA1锥体细胞中引起新的树突棘和突触。与树突棘和突触密度平行,雌二醇使NMDA受体结合增加46%。雌二醇还提高了CA1锥体细胞对NMDA受体介导的突触输入的敏感性,这种效应与雌二醇诱导的这些细胞顶端树突树中树突棘密度的增加相关[1]。雌二醇在急性解离和培养的新纹状体神经元中通过Ca2 +通道可逆地减少Ba2 +进入。雌二醇也可降低Ba2 +电流,但在大鼠新纹状体神经元中的效果明显低于雌二醇[2]。雌二醇剂量依赖性地抑制IL-1,TNF-α和IL-1以及TNF诱导的生物可测定的IL-6的产生。雌二醇阻断TNF诱导的IL-6产生和来自新生小鼠颅骨的原代骨细胞培养物中的破骨细胞发育[3]。 |
| 体内研究 | 雌二醇在成年大鼠动情周期中在海马突触密度中起作用[4]。雌二醇可逆转卵巢切除引起的脊柱密度降低。雌二醇与黄体酮联合使用可提高脊柱密度2至6小时,但单独使用雌二醇可降低脊柱密度[5]。 |
| 细胞实验 | 简而言之,用一系列稀释的上清液培养B9细胞(5×103 /孔的96孔板),最终体积为100mL RPMI 1640,补充有5×10-5mol / L的2-巯基乙醇。在平底微量滴定板中,10%FBS,100U / mL青霉素和100μg/ mL链霉素。 42小时后,加入0.5μCi的[3H]胸苷。 6小时后,收获细胞并测定掺入的放射性。从用已知量的重组人或小鼠IL-6建立的标准曲线定量IL-6。抗IL-6单克隆抗体完全抑制B9细胞响应重组小鼠IL-6而增殖的能力。此外,我们确定B9细胞不响应IL-1或TNF而增殖,并且针对这两种细胞因子的抗体不响应IL-6对细胞增殖的影响。此外,确定雌二醇(17β-雌二醇)和在这些实验中用作载体的0.01%乙醇都不会对生物测定产生任何影响。 |
| 动物实验 | 将成年雌性Sprague Dawley大鼠圈养在12小时光照/黑暗循环中,无限制地获取食物和水。将这些动物保持性腺完整(完整),卵巢切除并用苯甲酸雌二醇(OVX + E)处理,或切除卵形并用芝麻油载体(OVX + O)处理。先前已经显示在这些实验中使用的激素治疗方案导致CA1锥体细胞树突棘和突触密度的差异。在电生理学分析前6天,使用无菌外科手术在Metofane麻醉下对动物进行卵巢切除术。手术后,将动物单独圈养。在手术后第3天和第4天,在100μL芝麻油载体中向OSX注射(sc)10μg17β-雌二醇苯甲酸酯;每次注射时OVX + O动物接收油类载体。在第二次雌二醇或载体注射后48小时,通过断头处死动物并从其脑中制备海马切片。性腺完整的动物在动情周期的随机阶段被杀死。 |
| 参考文献 |
| 密度 | 1.2±0.1 g/cm3 |
|---|---|
| 沸点 | 445.9±45.0 °C at 760 mmHg |
| 熔点 | 173ºC |
| 分子式 | C18H24O2 |
| 分子量 | 272.382 |
| 闪点 | 209.6±23.3 °C |
| 精确质量 | 272.177643 |
| PSA | 40.46000 |
| LogP | 4.13 |
| 外观性状 | 白色至灰白色结晶粉末 |
| 蒸汽压 | 0.0±1.1 mmHg at 25°C |
| 折射率 | 1.599 |
| 储存条件 | 本品密封避光干燥保存。 |
| 稳定性 | 雌激素的作用,在未成年女子能促使第二性征的发育和性器官的最后形成。在成年女子,除保持上述第二性征外,并能使子宫内膜发生一系列的变化,产生周期性月经。也能增加子宫和输卵管的活动,并增加子宫肌对催产素的敏感性。此外,雌激素还能增加骨骼钙盐的沉积,加速骨骺闭合,大剂量时能升高血清甘油三脂和磷脂,引起水钠潴留。较大剂量时雌激素还能作用下丘脑-垂体系统,抑制促性腺激素及催乳素的分泌,并能抵消雄激素的主要作用。 |
| 水溶解性 | 水溶性:不溶;水溶解度:3.6 mg/l 27 °C;易溶于:二噁烷,二甲基甲酰胺;可溶于:丙酮;微溶:乙醇;极微溶:乙醚 |
| 分子结构 | 1、 摩尔折射率:79.50 2、 摩尔体积(cm3/mol):232.6 3、 等张比容(90.2K):615.4 4、 表面张力(dyne/cm):48.9 5、 极化率(10-24cm3):31.51 |
| 计算化学 | 1.疏水参数计算参考值(XlogP):无 2.氢键供体数量:2 3.氢键受体数量:2 4.可旋转化学键数量:0 5.互变异构体数量:9 6.拓扑分子极性表面积40.5 7.重原子数量:20 8.表面电荷:0 9.复杂度:382 10.同位素原子数量:0 11.确定原子立构中心数量:5 12.不确定原子立构中心数量:0 13.确定化学键立构中心数量:0 14.不确定化学键立构中心数量:0 15.共价键单元数量:1 |
| 更多 | 1. 性状:白色或乳白色叶片状或针状结晶(乙醇溶液);无臭。在空气中稳定 2. 密度(g/mL,25/4℃):未确定 3. 相对蒸汽密度(g/mL,空气=1):未确定 4. 熔点(ºC):178-179°C(lit.) 5. 沸点(ºC,常压):未确定 6. 沸点(ºC, 5.2 kPa):未确定 7. 折射率:80.4 ° (C=1, Dioxane) 8. 闪点(ºC):未确定 9. 比旋光度(º):未确定 10. 自燃点或引燃温度(ºC):未确定 11. 蒸气压(kPa,25 ºC):未确定 12. 饱和蒸气压(kPa,60 ºC):未确定 13. 燃烧热(KJ/mol):未确定 14. 临界温度(ºC):未确定 15. 临界压力(KPa):未确定 16. 油水(辛醇/水)分配系数的对数值:未确定 17. 爆炸上限(%,V/V):未确定 18. 爆炸下限(%,V/V):未确定 19. 溶解性:易溶于乙醇,溶于丙酮、氯仿、二氧六环和碱溶液,微溶于植物油,几乎不溶于水 |
|
模块1. 化学品 1.1 产品标识符 : β-雌二醇 产品名称 1.2 鉴别的其他方法 3,17β-Dihydroxy-1,3,5(10)-estratriene 1,3,5-Estratriene-3,17β-diol
Dihydrofolliculin 17β-Estradiol 1.3 有关的确定了的物质或混合物的用途和建议不适合的用途 仅用于研发。不作为药品、家庭或其它用途。 模块2. 危险性概述 2.1 GHS-分类 致癌性 (类别 2) 生殖毒性 (类别 1A) 影响哺乳或通过哺乳产生影响 2.2 GHS 标记要素,包括预防性的陈述 象形图 警示词危险 危险申明 H351怀疑会致癌。 H360可能对生育能力或胎儿造成伤害。 H362可能对母乳喂养的儿童造成伤害。 警告申明 预防措施 P201在使用前获取特别指示。 P202在读懂所有安全防范措施之前切勿操作。 P260不要吸入粉尘或烟雾。 P263怀孕/ 哺乳期间避免接触。 P264操作后彻底清洁皮肤。 P270使用本产品时不要进食、饮水或吸烟。 P281使用所需的个人防护设备。 事故响应 P308 + P313如接触到或有疑虑:求医/ 就诊。 安全储存 P405存放处须加锁。 废弃处置 P501将内容物/ 容器处理到得到批准的废物处理厂。 只限于专业使用者。 2.3 其它危害物 - 无 模块3. 成分/组成信息 3.1 物 质 : 3,17β-Dihydroxy-1,3,5(10)-estratriene 别名 1,3,5-Estratriene-3,17β-diol Dihydrofolliculin 17β-Estradiol : C18H24O2 分子式 : 272.38 g/mol 分子量 组分浓度或浓度范围 Estradiol <=100% 化学文摘登记号(CAS50-28-2 No.)200-023-8 EC-编号 模块4. 急救措施 4.1 必要的急救措施描述 一般的建议 请教医生。 向到现场的医生出示此安全技术说明书。 吸入 如果吸入,请将患者移到新鲜空气处。 如呼吸停止,进行人工呼吸。 请教医生。 皮肤接触 用肥皂和大量的水冲洗。 请教医生。 眼睛接触 用水冲洗眼睛作为预防措施。 食入 切勿给失去知觉者通过口喂任何东西。 用水漱口。 请教医生。 4.2 主要症状和影响,急性和迟发效应 据我们所知,此化学,物理和毒性性质尚未经完整的研究。 4.3 及时的医疗处理和所需的特殊处理的说明和指示 无数据资料 模块5. 消防措施 5.1 灭火介质 灭火方法及灭火剂 用水雾,抗乙醇泡沫,干粉或二氧化碳灭火。 5.2 源于此物质或混合物的特别的危害 碳氧化物 5.3 给消防员的建议 如必要的话,戴自给式呼吸器去救火。 5.4 进一步信息 无数据资料 模块6. 泄露应急处理 6.1 作业人员防护措施、防护装备和应急处置程序 使用个人防护用品。 避免粉尘生成。 避免吸入蒸气、烟雾或气体。 保证充分的通风。 人员疏散到安全区域。 避免吸入粉尘。 6.2 环境保护措施 如能确保安全,可采取措施防止进一步的泄漏或溢出。 不要让产品进入下水道。 6.3 泄漏化学品的收容、清除方法及所使用的处置材料 收集和处置时不要产生粉尘。 扫掉和铲掉。 放入合适的封闭的容器中待处理。 6.4 参考其他部分 丢弃处理请参阅第13节。 模块7. 操作处置与储存 7.1 安全操作的注意事项 避免曝露:使用前需要获得专门的指导。避免接触皮肤和眼睛。 避免形成粉尘和气溶胶。 在有粉尘生成的地方,提供合适的排风设备。 7.2 安全储存的条件,包括任何不兼容性 贮存在阴凉处。 使容器保持密闭,储存在干燥通风处。 7.3 特定用途 无数据资料 模块8. 接触控制和个体防护 8.1 容许浓度 最高容许浓度 没有已知的国家规定的暴露极限。 8.2 暴露控制 适当的技术控制 根据良好的工业卫生和安全规范进行操作。 休息前和工作结束时洗手。 个体防护设备 眼/面保护 带有防护边罩的安全眼镜符合 EN166要求请使用经官方标准如NIOSH (美国) 或 EN 166(欧盟) 检测与批准的设备防护眼部。 皮肤保护 戴手套取 手套在使用前必须受检查。 请使用合适的方法脱除手套(不要接触手套外部表面),避免任何皮肤部位接触此产品. 使用后请将被污染过的手套根据相关法律法规和有效的实验室规章程序谨慎处理. 请清洗并吹干双手 所选择的保护手套必须符合EU的89/686/EEC规定和从它衍生出来的EN 376标准。 完全接触 物料: 丁腈橡胶 最小的层厚度 0.11 mm 溶剂渗透时间: 480 min 测试过的物质Dermatril® (KCL 740 / Z677272, 规格 M) 飞溅保护 物料: 丁腈橡胶 最小的层厚度 0.11 mm 溶剂渗透时间: 480 min 测试过的物质Dermatril® (KCL 740 / Z677272, 规格 M) , 测试方法 EN374 如果以溶剂形式应用或与其它物质混合应用,或在不同于EN 374规定的条件下应用,请与EC批准的手套的供应商联系。 这个推荐只是建议性的,并且务必让熟悉我们客户计划使用的特定情况的工业卫生学专家评估确认才可. 这不应该解释为在提供对任何特定使用情况方法的批准. 身体保护 防渗透的衣服, 防护设备的类型必须根据特定工作场所中的危险物的浓度和数量来选择。 呼吸系统防护 如危险性评测显示需要使用空气净化的防毒面具,请使用全面罩式多功能微粒防毒面具N100型(US )或P3型(EN 143)防毒面具筒作为工程控制的候补。如果防毒面具是保护的唯一方式,则使用全面罩式送风防毒 面具。 呼吸器使用经过测试并通过政府标准如NIOSH(US)或CEN(EU)的呼吸器和零件。 模块9. 理化特性 9.1 基本的理化特性的信息 a) 外观与性状 形状: 粉末 颜色: 白色 b) 气味 无数据资料 c) 气味阈值 无数据资料 d) pH值 无数据资料 e) 熔点/凝固点 熔点/凝固点: 176 - 180 °C - lit. f) 沸点、初沸点和沸程 无数据资料 g) 闪点 无数据资料 h) 蒸发速率 无数据资料 i) 易燃性(固体,气体) 无数据资料 j) 高的/低的燃烧性或爆炸性限度 无数据资料 k) 蒸气压 无数据资料 l) 蒸汽密度 无数据资料 m) 密度/相对密度 无数据资料 n) 水溶性 无数据资料 o) n-辛醇/水分配系数 无数据资料 p) 自燃温度 无数据资料 q) 分解温度 无数据资料 r) 粘度 无数据资料 模块10. 稳定性和反应活性 10.1 反应性 无数据资料 10.2 稳定性 无数据资料 10.3 危险反应 无数据资料 10.4 应避免的条件 无数据资料 10.5 不相容的物质 强氧化剂 10.6 危险的分解产物 其它分解产物 - 无数据资料 模块11. 毒理学资料 11.1 毒理学影响的信息 急性毒性 无数据资料 皮肤刺激或腐蚀 无数据资料 眼睛刺激或腐蚀 无数据资料 呼吸道或皮肤过敏 无数据资料 生殖细胞致突变性 细胞突变性-体外试验 - 大鼠 - 其他细胞类型 DNA损伤 细胞突变性-体内试验 - 大鼠 - 经口 形态变形 致癌性 美国国家毒理学研究计划(关于致癌物的十号报告)已经确认固醇类雌激素为人类致癌物,因为大量证据表明 ,接触固醇类同人类肿瘤发生间存在因果关系。 该产品是或包含被IARC, ACGIH, EPA, 和 NTP 列为致癌物的组分 可疑人类致癌物 IARC: 此产品中没有大于或等于 0。1%含量的组分被 IARC鉴别为可能的或肯定的人类致癌物。 生殖毒性 可疑人类的生殖毒物 对胎儿的损害不能排除。 已知的人类生殖毒物 影响哺乳或通过哺乳产生影响 有可能损害生育能力。有损害胎儿的嫌疑。 特异性靶器官系统毒性(一次接触) 无数据资料 特异性靶器官系统毒性(反复接触) 无数据资料 吸入危险 无数据资料 潜在的健康影响 吸入吸入可能有害。 可能引起呼吸道刺激。 摄入如服入是有害的。 皮肤通过皮肤吸收可能有害。 可能引起皮肤刺激。 眼睛可能引起眼睛刺激。 接触后的征兆和症状 据我们所知,此化学,物理和毒性性质尚未经完整的研究。 附加说明 化学物质毒性作用登记: KG2975000 模块12. 生态学资料 12.1 生态毒性 对鱼类的毒性半数致死浓度(LC50) - 虹鳟 (红鳟鱼) - > 0.5 mg/l - 96 h 12.2 持久性和降解性 无数据资料 12.3 潜在的生物累积性 无数据资料 12.4 土壤中的迁移性 无数据资料 12.5 PBT 和 vPvB的结果评价 无数据资料 12.6 其它不良影响 模块13. 废弃处置 13.1 废物处理方法 产品 将剩余的和不可回收的溶液交给有许可证的公司处理。 与易燃溶剂相溶或者相混合,在备有燃烧后处理和洗刷作用的化学焚化炉中燃烧 受污染的容器和包装 按未用产品处置。 模块14. 运输信息 14.1 联合国危险货物编号 欧洲陆运危规: -国际海运危规: -国际空运危规: - 14.2 联合国运输名称 欧洲陆运危规: 非危险货物 国际海运危规: 非危险货物 国际空运危规: 非危险货物 14.3 运输危险类别 欧洲陆运危规: -国际海运危规: -国际空运危规: - 14.4 包裹组 欧洲陆运危规: -国际海运危规: -国际空运危规: - 14.5 环境危险 欧洲陆运危规: 否国际海运危规国际空运危规: 否 海洋污染物(是/否): 否 14.6 对使用者的特别提醒 无数据资料 模块 15 - 法规信息 N/A 模块16 - 其他信息 N/A |
|
雌二醇毒理学数据: 急性毒性: 主要的刺激性影响: 在皮肤上面:刺激皮肤和黏膜 在眼睛上面:刺激的影响。 致敏作用:没有已知的敏化影响。 雌二醇生态学数据: 总括注解 水危害级别3(德国规例)(通过名单进行自我评估)该物质对水有极其危害的。 即使是小量不要让该产品接触地下水、水道或污水系统。 即使是极其小量的产品渗入地下也会对饮用水造成危险 若无政府许可,勿将材料排入周围环境。 |
| 符号 |

GHS08 |
|---|---|
| 信号词 | Danger |
| 危害声明 | H351-H360Fd-H362 |
| 警示性声明 | P201-P263-P281-P308 + P313 |
| 个人防护装备 | Eyeshields;full-face particle respirator type N100 (US);Gloves;respirator cartridge type N100 (US);type P1 (EN143) respirator filter;type P3 (EN 143) respirator cartridges |
| 危害码 (欧洲) | T:Toxic |
| 风险声明 (欧洲) | R45 |
| 安全声明 (欧洲) | S53-S45 |
| 危险品运输编码 | 2811 |
| WGK德国 | 3 |
| RTECS号 | KG2975000 |
| 包装等级 | III |
| 危险类别 | 6.1 |
| 雌二醇上游产品 9 | |
|---|---|
| 雌二醇下游产品 10 | |
1.用雄甾-4-烯-19-去甲基-3,17-二酮经溴化在2-和6-位引入溴,然后在4-乙基-2-甲基吡啶存在下脱溴化氢(消除),生成2,4,6-雄甾三烯-19-去甲基-3,17-二酮,再经高温裂解生成雌酚酮,进而锂铝氢还原制得该品。
|
Inducible, tightly regulated and growth condition-independent transcription factor in Saccharomyces cerevisiae.
Nucleic Acids Res. 42(17) , e130, (2014) The precise control of gene expression is essential in basic biological research as well as in biotechnological applications. Most regulated systems available in yeast enable only the overexpression o... |
|
|
A precisely substituted benzopyran targets androgen refractory prostate cancer cells through selective modulation of estrogen receptors.
Toxicol. Appl. Pharmacol. 283(3) , 187-97, (2015) Dietary consumption of phytoestrogens like genistein has been linked with lower incidence of prostate cancer. The estradiol-like benzopyran core of genistein confers estrogen receptor-β (ER-β) selecti... |
|
|
Alpha-fetoprotein, identified as a novel marker for the antioxidant effect of placental extract, exhibits synergistic antioxidant activity in the presence of estradiol.
PLoS ONE 9(6) , e99421, (2014) Placenta, as a reservoir of nutrients, has been widely used in medical and cosmetic materials. Here, we focused on the antioxidant properties of placental extract and attempted to isolate and identify... |
| 17b-OH-estradiol |
| Estradiol-17β |
| 17b-Estradiol |
| Epiestriol 50 |
| Estra-1,3,5(10)-triene-3,17-diol, (17β)- |
| 17β estradiol |
| 17-Beta-Estradiol |
| δ-Estradiol |
| Destradiol |
| Estradiol-17 β |
| Oestra-1,3,5(10)-triene-3,17β-diol |
| 17b-Oestra-1,3,5(10)-triene-3,17-diol |
| Estra-1(10),2,4-triene-3,17-diol, (17β)- |
| δ-Oestradiol |
| estra-1(10),2,4-triene-3,17-diol, (17b)- |
| Oestradiol-17b |
| 3,17β-Dihydroxyestra-1,3,5(10)-triene |
| D-3,17b-Estradiol |
| estradiol 17b |
| Ovocylin |
| (17b)-Estra-1,3,5(10)-triene-3,17-diol |
| 3,17β-Estradiol |
| 3,17b-Dihydroxyestra-1,3,5(10)-triene |
| 17-β-estradiol |
| 3,17b-Oestradiol |
| Oestrogel |
| B-Estradiol |
| Estradiol-17-β |
| 17-β-OH-estradiol |
| D-3,17b-Oestradiol |
| Estrogel |
| Oestra-1,3,5(10)-triene-3,17b-diol |
| Estradiolum |
| 3,17b-Dihydroxy-1,3,5(10)-oestratriene |
| Estradiol |
| Estradiol, β- |
| Estradiol-17b |
| Estra-1,3,5(10)-triene-3,17-diol(17b)- |
| 17b-Oestradiol |
| (+)-3,17b-Estradiol |
| 17β-estradiol |
| (17β)-Estra-1(10),2,4-triene-3,17-diol |
| Estradiolo [DCIT] |
| 17beta-estradiol |
| 17β estradiol (E2) |
| D-3,17β-Estradiol |
| 17b-OH-oestradiol |
| 17β-OH-oestradiol |
| 17-β-OH-oestradiol |
| 13β-Methyl-1,3,5(10)-gonatriene-3,17β-ol |
| Estra-1,3,5(10)-triene-3,17b-diol |
| 3,17b-Estradiol |
| Oestradiol-17β |
| FEMPATCH |
| MFCD01074033 |
| (8R,9S,13S,14S,17S)-13-Methyl-7,8,9,11,12,13,14,15,16,17-decahydro-6H-cyclopenta[a]phenanthren-3,17-diol |
| 13b-Methyl-1,3,5(10)-gonatriene-3,17b-ol |
| EINECS 200-023-8 |
| Estracomb TTS |
| Estradiol-3,17b |
| (17b)-estra-1(10),2,4-triene-3,17-diol |
| Profoliol B |
| (+)-3,17β-Estradiol |
| 17β-OH-estradiol |
| 17β-Oestradiol |
| β-estradiol |

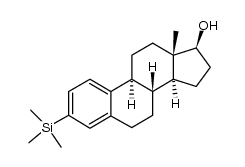 CAS号250331-30-7
CAS号250331-30-7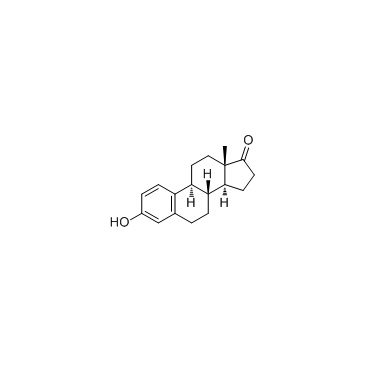 CAS号53-16-7
CAS号53-16-7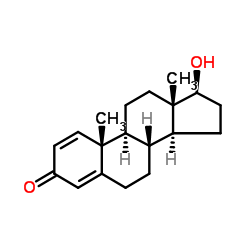 CAS号846-48-0
CAS号846-48-0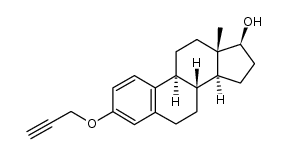 CAS号28151-61-3
CAS号28151-61-3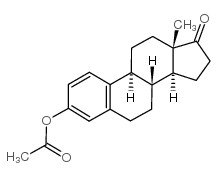 CAS号901-93-9
CAS号901-93-9 CAS号14982-15-1
CAS号14982-15-1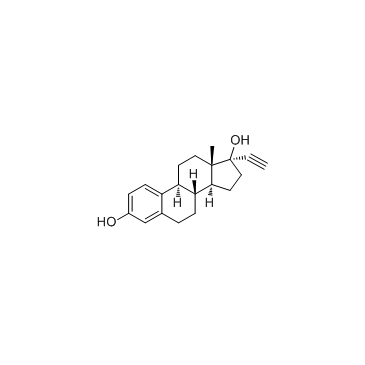 CAS号57-63-6
CAS号57-63-6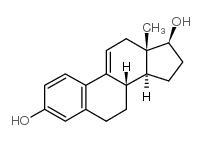 CAS号791-69-5
CAS号791-69-5 CAS号69455-04-5
CAS号69455-04-5 CAS号1089-78-7
CAS号1089-78-7 CAS号5976-61-4
CAS号5976-61-4 CAS号362-05-0
CAS号362-05-0 CAS号50-27-1
CAS号50-27-1 CAS号3571-53-7
CAS号3571-53-7 CAS号3758-34-7
CAS号3758-34-7 CAS号362-07-2
CAS号362-07-2 CAS号313-06-4
CAS号313-06-4 CAS号14364-66-0
CAS号14364-66-0 CAS号120858-07-3
CAS号120858-07-3
