Cyclopentenone

Cyclopentenone structure
|
Common Name | Cyclopentenone | ||
|---|---|---|---|---|
| CAS Number | 930-30-3 | Molecular Weight | 82.10 | |
| Density | 1.0±0.1 g/cm3 | Boiling Point | 136.0±0.0 °C at 760 mmHg | |
| Molecular Formula | C5H6O | Melting Point | N/A | |
| MSDS | Chinese USA | Flash Point | 42.2±0.0 °C | |
| Symbol |

GHS02 |
Signal Word | Warning | |
Use of CyclopentenoneCyclopent-2-enone is a biochemical reagent that can be used as a biological material or organic compound for life science related research. |
| Name | 2-Cyclopenten-1-one |
|---|---|
| Synonym | More Synonyms |
| Description | Cyclopent-2-enone is a biochemical reagent that can be used as a biological material or organic compound for life science related research. |
|---|---|
| Related Catalog |
| Density | 1.0±0.1 g/cm3 |
|---|---|
| Boiling Point | 136.0±0.0 °C at 760 mmHg |
| Molecular Formula | C5H6O |
| Molecular Weight | 82.10 |
| Flash Point | 42.2±0.0 °C |
| Exact Mass | 82.041862 |
| PSA | 17.07000 |
| LogP | -0.05 |
| Vapour Pressure | 7.5±0.2 mmHg at 25°C |
| Index of Refraction | 1.492 |
| InChIKey | BZKFMUIJRXWWQK-UHFFFAOYSA-N |
| SMILES | O=C1C=CCC1 |
| Storage condition | 2-8°C |
| Water Solubility | almost insoluble |
| Symbol |

GHS02 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H226 |
| Personal Protective Equipment | Eyeshields;Faceshields;full-face respirator (US);Gloves;multi-purpose combination respirator cartridge (US);type ABEK (EN14387) respirator filter |
| Hazard Codes | Xn:Harmful; |
| Risk Phrases | R10;R22;R36/37/38 |
| Safety Phrases | S16-S36-S36/37/39 |
| RIDADR | UN 1224 3/PG 3 |
| WGK Germany | 3 |
| Packaging Group | III |
| Hazard Class | 3 |
| HS Code | 2914299000 |
| Precursor 0 | |
|---|---|
| DownStream 10 | |
| HS Code | 2914299000 |
|---|---|
| Summary | 2914299000. other cyclanic, cyclenic or cyclotherpenic ketones without other oxygen function. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:5.5%. General tariff:30.0% |
|
The cyclopentenone prostaglandin 15d-PGJ2 inhibits the NLRP1 and NLRP3 inflammasomes.
J. Immunol. 194(6) , 2776-85, (2015) Inflammasomes are cytosolic protein complexes that respond to diverse danger signals by activating caspase-1. The sensor components of the inflammasome, often proteins of the nucleotide-binding oligom... |
|
|
Enantioselective synthesis of 4-heterosubstituted cyclopentenones.
J. Org. Chem. 78(8) , 4202-6, (2013) Racemic 4-hydroxycyclopentenone, readily derived from furfuryl alcohol, can be transformed via its O-Boc derivative to 4-acyloxy, 4-aryloxy-, 4-amino-, or 4-thio-substituted cyclopentenones with high ... |
|
|
Sinularones A-I, new cyclopentenone and butenolide derivatives from a marine soft coral Sinularia sp. and their antifouling activity.
Mar. Drugs 10(6) , 1331-44, (2012) Nine new compounds, namely sinularones A-I (1-9), characterized as cyclopentenone and butenolide-type analogues, were isolated from a soft coral Sinularia sp., together with a known butenolide (10). T... |
| 2-Cyclopentenone |
| cyclopent-2-en-1-one |
| EINECS 213-213-0 |
| 3-Cyclopenten-2-one |
| Cyclopentenone |
| MFCD00001401 |
| 2-Cyclopenten-1-one |
 CAS#:110027-73-1
CAS#:110027-73-1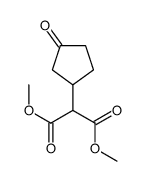 CAS#:111209-96-2
CAS#:111209-96-2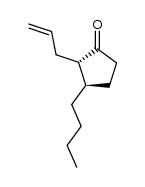 CAS#:109900-41-6
CAS#:109900-41-6 CAS#:109900-42-7
CAS#:109900-42-7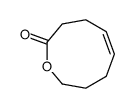 CAS#:110189-60-1
CAS#:110189-60-1![3-[dimethyl(phenyl)silyl]cyclopentan-1-one structure](https://image.chemsrc.com/caspic/417/109023-10-1.png) CAS#:109023-10-1
CAS#:109023-10-1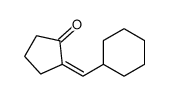 CAS#:106115-44-0
CAS#:106115-44-0 CAS#:39627-61-7
CAS#:39627-61-7 CAS#:395-23-3
CAS#:395-23-3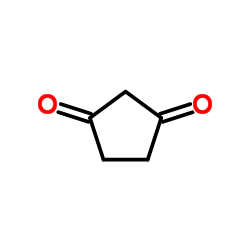 CAS#:3859-41-4
CAS#:3859-41-4
