omeprazole

omeprazole structure
|
Common Name | omeprazole | ||
|---|---|---|---|---|
| CAS Number | 73590-58-6 | Molecular Weight | 345.416 | |
| Density | 1.4±0.1 g/cm3 | Boiling Point | 600.0±60.0 °C at 760 mmHg | |
| Molecular Formula | C17H19N3O3S | Melting Point | 156ºC | |
| MSDS | Chinese USA | Flash Point | 316.7±32.9 °C | |
| Symbol |

GHS07 |
Signal Word | Warning | |
Use of omeprazoleOmeprazole(Prilosec) is a proton pump inhibitor used in the treatment of dyspepsia.Target: Proton PumpOmeprazole is a proton pump inhibitor used in the treatment of dyspepsia, peptic ulcer disease, gastroesophageal reflux disease, laryngopharyngeal reflux, and Zollinger-Ellison syndrome. Omeprazole virtually eliminated intragastric acidity in all patients: the median 24 hour intragastric pH rose from 1.4 to 5.3 and the mean hourly hydrogen ion activity fell from 38.50 to 1.95 mmol(mEq)/1 (p less than 0.001). This inhibition of 24 hour intragastric acidity is more profound than that previously reported with either cimetidine 1 g daily or ranitidine 300 mg daily [1]. The pharmacokinetics of omeprazole were studied in a group of healthy male subjects after single and repeated oral doses of 30 and 60 mg. Absorption of omeprazole from its enteric-coated formulation was unpredictable. There was a highly significant increase in the area under the plasma concentration time curve (AUC) after repeated dosing. Omeprazole increases its own relative availability following repeated dosing. This may be due to inhibition of gastric acid secretion by omeprazole which is an acid-labile compound [2].Clinical indications: Duodenal ulcer; Endocrine tumor; Esophagitis; Gastroesophageal reflux; Helicobacter pylori infection; Stomach ulcer; Zollinger-Ellison syndromeToxicity: Symptoms of overdose include confusion, drowsiness, blurred vision, tachycardia, nausea, diaphoresis, flushing, headache, and dry mouth. |
| Name | 5-methoxy-2-{[(4-methoxy-3,5-dimethylpyridin-2-yl)methyl]sulfinyl}-1H-benzimidazole |
|---|---|
| Synonym | More Synonyms |
| Description | Omeprazole(Prilosec) is a proton pump inhibitor used in the treatment of dyspepsia.Target: Proton PumpOmeprazole is a proton pump inhibitor used in the treatment of dyspepsia, peptic ulcer disease, gastroesophageal reflux disease, laryngopharyngeal reflux, and Zollinger-Ellison syndrome. Omeprazole virtually eliminated intragastric acidity in all patients: the median 24 hour intragastric pH rose from 1.4 to 5.3 and the mean hourly hydrogen ion activity fell from 38.50 to 1.95 mmol(mEq)/1 (p less than 0.001). This inhibition of 24 hour intragastric acidity is more profound than that previously reported with either cimetidine 1 g daily or ranitidine 300 mg daily [1]. The pharmacokinetics of omeprazole were studied in a group of healthy male subjects after single and repeated oral doses of 30 and 60 mg. Absorption of omeprazole from its enteric-coated formulation was unpredictable. There was a highly significant increase in the area under the plasma concentration time curve (AUC) after repeated dosing. Omeprazole increases its own relative availability following repeated dosing. This may be due to inhibition of gastric acid secretion by omeprazole which is an acid-labile compound [2].Clinical indications: Duodenal ulcer; Endocrine tumor; Esophagitis; Gastroesophageal reflux; Helicobacter pylori infection; Stomach ulcer; Zollinger-Ellison syndromeToxicity: Symptoms of overdose include confusion, drowsiness, blurred vision, tachycardia, nausea, diaphoresis, flushing, headache, and dry mouth. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.4±0.1 g/cm3 |
|---|---|
| Boiling Point | 600.0±60.0 °C at 760 mmHg |
| Melting Point | 156ºC |
| Molecular Formula | C17H19N3O3S |
| Molecular Weight | 345.416 |
| Flash Point | 316.7±32.9 °C |
| Exact Mass | 345.114716 |
| PSA | 96.31000 |
| LogP | 2.17 |
| Vapour Pressure | 0.0±1.7 mmHg at 25°C |
| Index of Refraction | 1.669 |
| InChIKey | SUBDBMMJDZJVOS-UHFFFAOYSA-N |
| SMILES | COc1ccc2nc(S(=O)Cc3ncc(C)c(OC)c3C)[nH]c2c1 |
| Storage condition | 2-8°C |
| Stability | Stable, but hygroscopic and photosensitive. Incompatible with strong oxidizing agents. Store in the dark. |
| Water Solubility | H2O: 0.5 mg/mL |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
MUTATION DATA
|
| Symbol |

GHS07 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H315-H319-H335 |
| Precautionary Statements | P305 + P351 + P338 |
| Personal Protective Equipment | dust mask type N95 (US);Eyeshields;Gloves |
| Hazard Codes | Xi:Irritant; |
| Risk Phrases | R36/37/38 |
| Safety Phrases | S26-S36 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 2 |
| RTECS | DD9087000 |
| Precursor 8 | |
|---|---|
| DownStream 7 | |
|
Novel Marmoset Cytochrome P450 2C19 in Livers Efficiently Metabolizes Human P450 2C9 and 2C19 Substrates, S-Warfarin, Tolbutamide, Flurbiprofen, and Omeprazole.
Drug Metab. Dispos. 43 , 1408-16, (2015) The common marmoset (Callithrix jacchus), a small New World monkey, has the potential for use in human drug development due to its evolutionary closeness to humans. Four novel cDNAs, encoding cytochro... |
|
|
HepG2 cells as an in vitro model for evaluation of cytochrome P450 induction by xenobiotics.
Arch. Pharm. Res. 38 , 691-704, (2015) Although various in vitro assays have been developed to evaluate the cytochrome P450 (CYP)-inducing potential of drug candidates, there is a continuing need for the development of a reliable model in ... |
|
|
Evaluation of the effect of TM208 on the activity of five cytochrome P450 enzymes using on-line solid-phase extraction HPLC-DAD: a cocktail approach.
J. Chromatogr. B. Analyt. Technol. Biomed. Life Sci. 923-924 , 29-36, (2013) A rapid, simple, and sensitive on-line solid-phase extraction HPLC-DAD method for simultaneous evaluation of the activity of five CYP450 isoforms (CYP1A2, CYP2C19, CYP2D6, CYP2E1 and CYP3A4) in vivo h... |
| 5-methoxy-2-[(4-methoxy-3,5-dimethyl-pyridin-2-yl)-methylsulfinyl]benzimidazole |
| 5-methoxy-2-[[(4-methoxy-3,5-dimethyl-2-pyridyl)methyl]sulfinyl]-1H-benzimidazole |
| Omepral |
| (RS)-5-Methoxy-2-(4-methoxy-3,5-dimethyl-2-pyridylmethylsulphinyl)benzimidazole |
| elgam |
| ANTRA |
| MEPRAL |
| Zoltum |
| 1H-Benzimidazole, 5-methoxy-2-(((4-methoxy-3,5-dimethyl-2-pyridinyl)methyl)sulfinyl)- |
| [14C]-Omeprazole |
| PRILOSEC |
| (-)-5-methoxy-2-[[(4-methoxy-3,5-dimethyl-2-pyridinyl)methyl]sulfinyl]-1H-benzimidazole |
| zimor |
| Losec |
| Omeprazen |
| Pepticum |
| prysma |
| 5-methoxy-2-{[(4-methoxy-3,5-dimethylpyridin-2-yl)methyl]sulfinyl}-1H-benzimidazole |
| EINECS 201-212-8 |
| (±)-Omeprazole |
| MFCD00083192 |
| Omeprazole |
| 5-methoxy-2-[[(4-methoxy-3,5-dimethyl-2-pyridinyl)-methyl]sulphinyl]-1H-benzimidazole |
| aulcer |
| 6-Methoxy-2-{[(4-methoxy-3,5-dimethylpyridin-2-yl)methyl]sulfinyl}-1H-benzimidazole |
| 5-Methoxy-2-{[(4-methoxy-3,5-dimethyl-2-pyridinyl)methyl]sulfinyl}-1H-benzimidazole |
| 1H-Benzimidazole, 5-methoxy-2-[[(4-methoxy-3,5-dimethyl-2-pyridinyl)methyl]sulfinyl]- |
| sulfonium, hydroxy(6-methoxy-1H-benzimidazol-2-yl)[(4-methoxy-3,5-dimethyl-2-pyridinyl)methyl]-, inner salt |
| ulcsep |
| 5-methoxy-2-[(4-methoxy-3,5-dimethyl-2-pyridyl)methylsulphinyl]-1H-benzimidazole |
| Miol |
| Gastrogard |
 CAS#:73590-85-9
CAS#:73590-85-9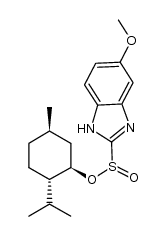 CAS#:1080503-61-2
CAS#:1080503-61-2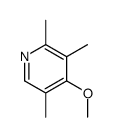 CAS#:109371-19-9
CAS#:109371-19-9![5-Methoxy-2-[[(4-methoxy-3,5-dimethyl-2-pyridinyl)methyl]thio]-1H-benzimidazole N-Oxide Structure](https://image.chemsrc.com/caspic/092/142885-92-5.png) CAS#:142885-92-5
CAS#:142885-92-5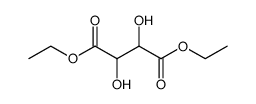 CAS#:408332-88-7
CAS#:408332-88-7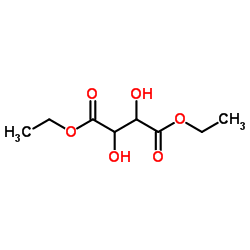 CAS#:13811-71-7
CAS#:13811-71-7 CAS#:861359-74-2
CAS#:861359-74-2 CAS#:26787-78-0
CAS#:26787-78-0 CAS#:37052-78-1
CAS#:37052-78-1 CAS#:60-54-8
CAS#:60-54-8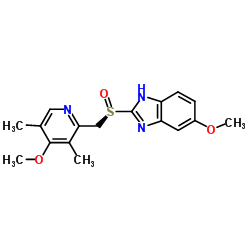 CAS#:119141-89-8
CAS#:119141-89-8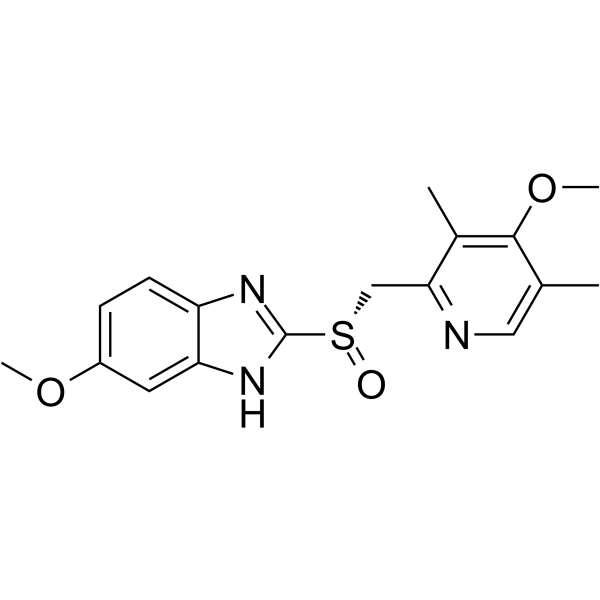 CAS#:119141-88-7
CAS#:119141-88-7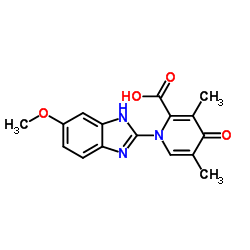 CAS#:1227380-90-6
CAS#:1227380-90-6
