(S)-Baclofen hydrochloride
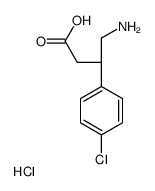
(S)-Baclofen hydrochloride structure
|
Common Name | (S)-Baclofen hydrochloride | ||
|---|---|---|---|---|
| CAS Number | 63701-56-4 | Molecular Weight | 250.12200 | |
| Density | N/A | Boiling Point | 364.3ºC at 760mmHg | |
| Molecular Formula | C10H13Cl2NO2 | Melting Point | 223 - 224 °C | |
| MSDS | USA | Flash Point | 174.1ºC | |
| Name | (3S)-4-amino-3-(4-chlorophenyl)butanoic acid,hydrochloride |
|---|---|
| Synonym | More Synonyms |
| Boiling Point | 364.3ºC at 760mmHg |
|---|---|
| Melting Point | 223 - 224 °C |
| Molecular Formula | C10H13Cl2NO2 |
| Molecular Weight | 250.12200 |
| Flash Point | 174.1ºC |
| Exact Mass | 249.03200 |
| PSA | 63.32000 |
| LogP | 3.35930 |
| InChIKey | WMNUVYYLMCMHLU-DDWIOCJRSA-N |
| SMILES | Cl.NCC(CC(=O)O)c1ccc(Cl)cc1 |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Personal Protective Equipment | Eyeshields;Gloves;type N95 (US);type P1 (EN143) respirator filter |
|---|---|
| RIDADR | UN 2811 6.1/PG 3 |
| RTECS | MW5084400 |
|
Effects of phaclofen and the enantiomers of baclofen on cardiovascular responses to intrathecal administration of L- and D-baclofen in the rat.
Eur. J. Pharmacol. 196 , 267, (1991) In a previous study it was found that i.t. administration of L-baclofen decreased arterial pressure and heart rate while D-baclofen differentially increased arterial pressure. The objective of the pre... |
|
|
Comparative stereostructure-activity studies on GABAA and GABAB receptor sites and GABA uptake using rat brain membrane preparations.
J. Neurochem. 47 , 898, (1986) The affinities of a number of analogues of gamma-aminobutyric acid (GABA) for GABAA and GABAB receptor sites and GABA uptake were studied using rat brain membrane preparations. Studies on the (S)-(+)-... |
|
|
3-(p-Chlorophenyl)-4-aminobutanoic acid--resolution into enantiomers and pharmacological activity.
Pol. J. Pharmacol. Pharm. 32 , 187, (1980) Racemic 3-(p-chlorophenyl)-4-aminobutanoic acid was resolved into enantiomers and their absolute configuration determined. Pharmacological activity of hydrochlorides of the racemic acid and its enanti... |
| UNII-799SU69U5P |
| S(-)-Baclofen hydrochloride |
| (-)-Baclofen hydrochloride |
| l-Baclofen hydrochloride |
| (-)-S-3-(p-Chlorophenyl)-4-aminobutanoic acid hydrochloride |
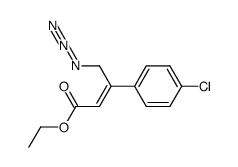 CAS#:537676-64-5
CAS#:537676-64-5 CAS#:1615-02-7
CAS#:1615-02-7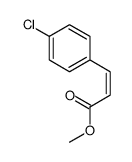 CAS#:20754-21-6
CAS#:20754-21-6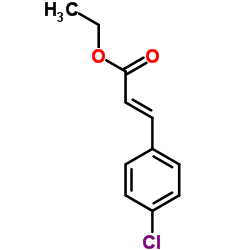 CAS#:24393-52-0
CAS#:24393-52-0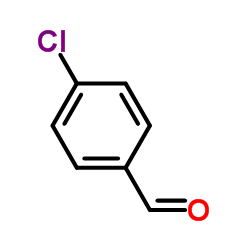 CAS#:104-88-1
CAS#:104-88-1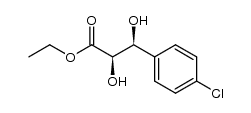 CAS#:1006386-90-8
CAS#:1006386-90-8