1-Methylindole
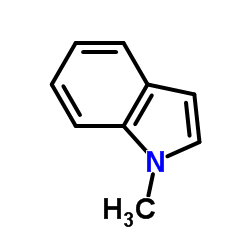
1-Methylindole structure
|
Common Name | 1-Methylindole | ||
|---|---|---|---|---|
| CAS Number | 603-76-9 | Molecular Weight | 131.17 | |
| Density | 1.0±0.1 g/cm3 | Boiling Point | 239.4±9.0 °C at 760 mmHg | |
| Molecular Formula | C9H9N | Melting Point | 94 - 95ºC | |
| MSDS | Chinese USA | Flash Point | 98.6±18.7 °C | |
Use of 1-Methylindole1-Methylindole is a biochemical reagent that can be used as a biological material or organic compound for life science related research. |
| Name | 1-Methylindole |
|---|---|
| Synonym | More Synonyms |
| Description | 1-Methylindole is a biochemical reagent that can be used as a biological material or organic compound for life science related research. |
|---|---|
| Related Catalog |
| Density | 1.0±0.1 g/cm3 |
|---|---|
| Boiling Point | 239.4±9.0 °C at 760 mmHg |
| Melting Point | 94 - 95ºC |
| Molecular Formula | C9H9N |
| Molecular Weight | 131.17 |
| Flash Point | 98.6±18.7 °C |
| Exact Mass | 131.073502 |
| PSA | 4.93000 |
| LogP | 2.54 |
| Vapour Pressure | 0.1±0.5 mmHg at 25°C |
| Index of Refraction | 1.570 |
| Storage condition | Refrigerator (+4°C) |
| Stability | Light sensitive. Combustible. Incompatible with strong oxidizing agents. |
| Water Solubility | insoluble |
| Personal Protective Equipment | Eyeshields;Gloves;half-mask respirator (US);multi-purpose combination respirator cartridge (US) |
|---|---|
| Hazard Codes | Xi:Irritant |
| Risk Phrases | R36/37/38 |
| Safety Phrases | S22-S24/25-S37/39-S26 |
| RIDADR | UN 3334 |
| WGK Germany | 3 |
| HS Code | 29339990 |
| Precursor 10 | |
|---|---|
| DownStream 10 | |
| HS Code | 2933990090 |
|---|---|
| Summary | 2933990090. heterocyclic compounds with nitrogen hetero-atom(s) only. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
|
Natural compounds boldine and menthol are antagonists of human 5-HT3 receptors: implications for treating gastrointestinal disorders.
Neurogastroenterol. Motil. 26(6) , 810-20, (2014) Impaired 5-HT3 receptor function is likely involved in the pathogenesis of functional gastrointestinal disorders (FGID) and 5-HT3 receptor antagonists are effective treatments for chemotherapy-induced... |
|
|
Degradation of substituted indoles by an indole-degrading methanogenic consortium.
Appl. Environ. Microbiol. 57(9) , 2622-7, (1991) Degradation of indole by an indole-degrading methanogenic consortium enriched from sewage sludge proceeded through a two-step hydroxylation pathway yielding oxindole and isatin. The ability of this co... |
|
|
Single-fluorophore-based fluorescent probes enable dual-channel detection of Ag⁺ and Hg²⁺ with high selectivity and sensitivity.
Anal. Chim. Acta 839 , 74-82, (2014) A new type of fluorescent probe capable of detecting Ag(+) and Hg(2+) in two independent channels was developed in the present work. Specifically, in CH3CN-MOPS mixed solvents with CH3CN/MOPS ratio (v... |
| indole, 1-methyl- |
| N-methylindole |
| 1H-Indole,1-methyl |
| 1-Methyl-1H-indole |
| methylindole |
| 1-methyl indole |
| 1-Methylindole |
| Indole, 1-methyl- (8CI) |
| MFCD00005800 |
| 1H-Indole, 1-methyl- |
| EINECS 210-057-5 |
| Indole,1-methyl |
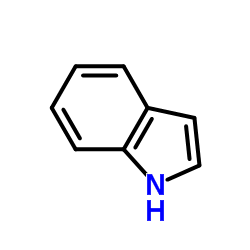 CAS#:120-72-9
CAS#:120-72-9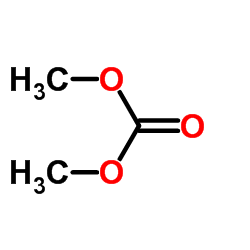 CAS#:616-38-6
CAS#:616-38-6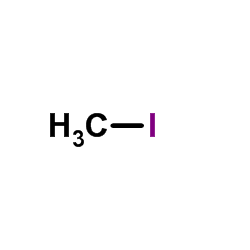 CAS#:74-88-4
CAS#:74-88-4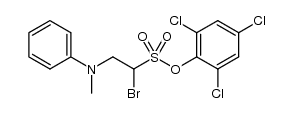 CAS#:1352621-88-5
CAS#:1352621-88-5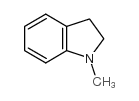 CAS#:824-21-5
CAS#:824-21-5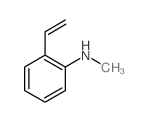 CAS#:5339-28-6
CAS#:5339-28-6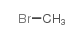 CAS#:74-83-9
CAS#:74-83-9 CAS#:1352334-73-6
CAS#:1352334-73-6 CAS#:81471-20-7
CAS#:81471-20-7 CAS#:16136-58-6
CAS#:16136-58-6 CAS#:1421259-27-9
CAS#:1421259-27-9 CAS#:105804-97-5
CAS#:105804-97-5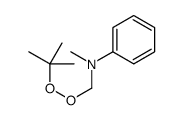 CAS#:52866-87-2
CAS#:52866-87-2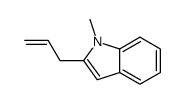 CAS#:111678-54-7
CAS#:111678-54-7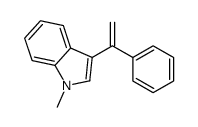 CAS#:112122-42-6
CAS#:112122-42-6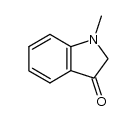 CAS#:3260-62-6
CAS#:3260-62-6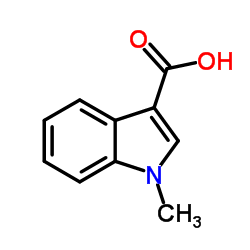 CAS#:32387-21-6
CAS#:32387-21-6 CAS#:151490-40-3
CAS#:151490-40-3 CAS#:6637-11-2
CAS#:6637-11-2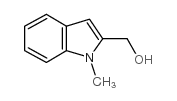 CAS#:1485-22-9
CAS#:1485-22-9
