Ingenol 3,20-dibenzoate
Modify Date: 2025-08-25 19:18:51
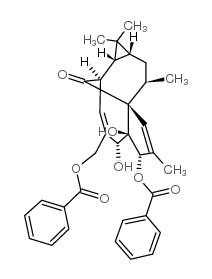
Ingenol 3,20-dibenzoate structure
|
Common Name | Ingenol 3,20-dibenzoate | ||
|---|---|---|---|---|
| CAS Number | 59086-90-7 | Molecular Weight | 556.64500 | |
| Density | 1.32 g/cm3 | Boiling Point | 697ºC at 760 mmHg | |
| Molecular Formula | C34H36O7 | Melting Point | N/A | |
| MSDS | N/A | Flash Point | 221.6ºC | |
Use of Ingenol 3,20-dibenzoateIngenol 3,20-dibenzoate is a potent protein kinase C (PKC) isoform-selective agonist. Ingenol 3,20-dibenzoate induces selective translocation of nPKC-delta, -epsilon, and -theta and PKC-mu from the cytosolic fraction to the particulate fraction and induces morphologically typical apoptosis through de novo synthesis of macromolecules. Ingenol 3,20-dibenzoate increases the IFN-γ production and degranulation by NK cells, especially when NK cells are stimulated by NSCLC cells[1][2]. |
| Name | ingenol 3,20-dibenzoate |
|---|---|
| Synonym | More Synonyms |
| Description | Ingenol 3,20-dibenzoate is a potent protein kinase C (PKC) isoform-selective agonist. Ingenol 3,20-dibenzoate induces selective translocation of nPKC-delta, -epsilon, and -theta and PKC-mu from the cytosolic fraction to the particulate fraction and induces morphologically typical apoptosis through de novo synthesis of macromolecules. Ingenol 3,20-dibenzoate increases the IFN-γ production and degranulation by NK cells, especially when NK cells are stimulated by NSCLC cells[1][2]. |
|---|---|
| Related Catalog | |
| In Vitro | Ingenol 3,20-dibenzoate (0-10000 nM; 4 hours) enhanced NK cells (stimulated by A549 cells and H1299 cells) degranulation[2]. Ingenol 3,20-dibenzoate (0.001-10 μg/ml) dose dependently promoted UT-7/EPO cell proliferation with an EC50 of 0.27 μg/ml (485 nM)[3]. |
| In Vivo | Ingenol 3,20-dibenzoate (20 μg/mouse; i.p.; on days 3, 7, and 11) lessens the anemia induced by 5-fluorouracil in an in vivo mouse model[3]. Animal Model: Male C57BL/6 mice (8–12 weeks old)[3] Dosage: 20 μg/mouse Administration: I.p.; On days 3, 7, and 11 Result: Severity of 5-FU–induced anemia was lessened by IDB treatment on days 12 and 20. |
| References |
| Density | 1.32 g/cm3 |
|---|---|
| Boiling Point | 697ºC at 760 mmHg |
| Molecular Formula | C34H36O7 |
| Molecular Weight | 556.64500 |
| Flash Point | 221.6ºC |
| Exact Mass | 556.24600 |
| PSA | 110.13000 |
| LogP | 4.54460 |
| Index of Refraction | 1.639 |
| Hazard Codes | Xn: Harmful; |
|---|---|
| Risk Phrases | R20/21/22 |
| Safety Phrases | 26-36 |
| ingenoldibenzoate |