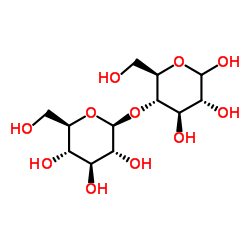
Maltitol
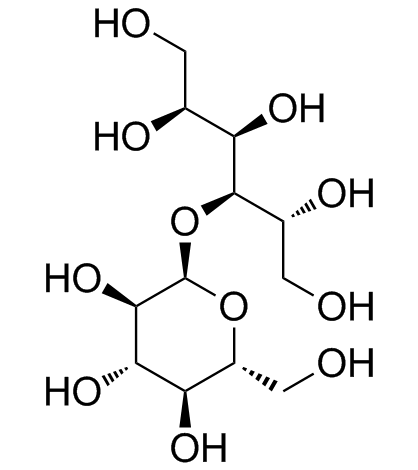
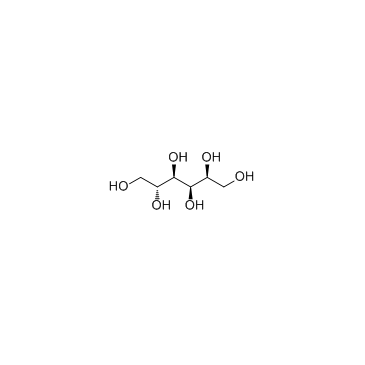
Maltitol structure
|
Common Name | Maltitol | ||
|---|---|---|---|---|
| CAS Number | 585-88-6 | Molecular Weight | 344.312 | |
| Density | 1.7±0.1 g/cm3 | Boiling Point | 788.5±60.0 °C at 760 mmHg | |
| Molecular Formula | C12H24O11 | Melting Point | 149-152 °C(lit.) | |
| MSDS | Chinese USA | Flash Point | 430.7±32.9 °C | |
Use of MaltitolMaltitol is a sugar alcohol used as a sugar substitute. It has 75-90% of the sweetness of sucrose (table sugar) and nearly identical properties. Maltitol may also be used as a plasticizer in gelatin capsules, as an emollient, and as a humectant[1]. |
| Name | maltitol |
|---|---|
| Synonym | More Synonyms |
| Description | Maltitol is a sugar alcohol used as a sugar substitute. It has 75-90% of the sweetness of sucrose (table sugar) and nearly identical properties. Maltitol may also be used as a plasticizer in gelatin capsules, as an emollient, and as a humectant[1]. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.7±0.1 g/cm3 |
|---|---|
| Boiling Point | 788.5±60.0 °C at 760 mmHg |
| Melting Point | 149-152 °C(lit.) |
| Molecular Formula | C12H24O11 |
| Molecular Weight | 344.312 |
| Flash Point | 430.7±32.9 °C |
| Exact Mass | 344.131866 |
| PSA | 200.53000 |
| LogP | -5.14 |
| Vapour Pressure | 0.0±6.2 mmHg at 25°C |
| Index of Refraction | 1.634 |
| Storage condition | 2-8°C |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
|
~% 
Maltitol CAS#:585-88-6 |
| Literature: Advanced Synthesis and Catalysis, , vol. 350, # 6 p. 829 - 836 |
|
~% 
Maltitol CAS#:585-88-6 |
| Literature: Journal of the American Chemical Society, , vol. 73, p. 4691 |
|
~%
Detail
|
| Literature: Carbohydrate Research, , vol. 173, p. 89 - 100 |
| Precursor 1 | |
|---|---|
| DownStream 0 | |
| HS Code | 2932999099 |
|---|---|
| Summary | 2932999099. other heterocyclic compounds with oxygen hetero-atom(s) only. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
|
Highly efficient extraction of anthocyanins from grape skin using deep eutectic solvents as green and tunable media.
Arch. Pharm. Res. 38 , 2143-52, (2015) Deep eutectic solvents (DESs) were investigated as tunable, environmentally benign, yet superior extraction media to enhance the extraction of anthocyanins from grape skin, which is usually discarded ... |
|
|
Preparation and chromatographic evaluation of a newly designed steviol glycoside modified-silica stationary phase in hydrophilic interaction liquid chromatography and reversed phase liquid chromatography.
J. Chromatogr. A. 1388 , 110-8, (2015) A diterpene glycoside compound, rebaudioside A (commonly abbreviated as RA), was immobilized onto porous silica surface through "thiol-ene" click chemistry strategy. The successful immobilization of t... |
|
|
Preparation of D-gulose from disaccharide lactitol using microbial and chemical methods.
Biosci. Biotechnol. Biochem. 77(2) , 253-8, (2013) When an M31 strain of Agrobacterium tumefaciens was grown in a mineral salt medium at 30 °C containing 1.0% lactitol as sole carbon source, a keto-sugar was efficiently accumulated in the supernatant.... |
| Maltisorb |
| maltimr |
| Maltit |
| Lesys |
| D-Glucitol, 4-O-α-D-glucopyranosyl- |
| amaltymr100 |
| D-4-O-α-D-Glucopyranosylglucitol |
| D-Maltitol |
| Malti Mr |
| Amalty P |
| Amalty Syrup |
| Glucitol, 4-O-α-D-glucopyranosyl-, D- |
| D-glucitol, 4-O-a-D-glucopyranosyl- |
| Malbit CH |
| Malbit CH 16385 |
| Maltitol |
| 4-O-a-D-glucopyranosyl-D-glucitol |
| Amalty MR 50 |
| D-4-O-a-D-Glucopyranosylglucitol |
| Malbit CR |
| MFCD00006600 |
| Amalty MR 100 |
| 4-O-a-D-glucopyranosyl- D-glucitol |
| δ-4-O-α-δ-Glucopyranosylglucitol |
| Amalty |
| 4-O-α-D-Glucopyranosyl-D-glucitol |
| δ-Maltitol |
| EINECS 209-567-0 |
| Amalty MR 20 |
| Mabit |
| D-Glucitol, 4--O-α-D-glucopyranosyl |
| Maltidex 100 |
| malbit |
| amaltisyrup |