N-Boc-Ethylenediamine

N-Boc-Ethylenediamine structure
|
Common Name | N-Boc-Ethylenediamine | ||
|---|---|---|---|---|
| CAS Number | 57260-73-8 | Molecular Weight | 160.214 | |
| Density | 1.0±0.1 g/cm3 | Boiling Point | 252.8±23.0 °C at 760 mmHg | |
| Molecular Formula | C7H16N2O2 | Melting Point | N/A | |
| MSDS | Chinese USA | Flash Point | 106.7±22.6 °C | |
| Symbol |

GHS05 |
Signal Word | Danger | |
Use of N-Boc-EthylenediaminePROTAC Linker 22 is a PROTAC linker, which refers to the alkyl chain composition. PROTAC Linker 22 can be used in the synthesis of a series of PROTACs. PROTACs contain two different ligands connected by a linker; one is a ligand for an E3 ubiquitin ligase and the other is for the target protein. PROTACs exploit the intracellular ubiquitin-proteasome system to selectively degrade target proteins[1]. |
| Name | N-Boc-Ethylenediamine |
|---|---|
| Synonym | More Synonyms |
| Description | PROTAC Linker 22 is a PROTAC linker, which refers to the alkyl chain composition. PROTAC Linker 22 can be used in the synthesis of a series of PROTACs. PROTACs contain two different ligands connected by a linker; one is a ligand for an E3 ubiquitin ligase and the other is for the target protein. PROTACs exploit the intracellular ubiquitin-proteasome system to selectively degrade target proteins[1]. |
|---|---|
| Related Catalog | |
| Target |
Alkyl-Chain |
| References |
| Density | 1.0±0.1 g/cm3 |
|---|---|
| Boiling Point | 252.8±23.0 °C at 760 mmHg |
| Molecular Formula | C7H16N2O2 |
| Molecular Weight | 160.214 |
| Flash Point | 106.7±22.6 °C |
| Exact Mass | 160.121185 |
| PSA | 64.35000 |
| LogP | 0.54 |
| Vapour Pressure | 0.0±0.5 mmHg at 25°C |
| Index of Refraction | 1.454 |
| InChIKey | AOCSUUGBCMTKJH-UHFFFAOYSA-N |
| SMILES | CC(C)(C)OC(=O)NCCN |
| Storage condition | Refrigerator (+4°C) |
| Symbol |

GHS05 |
|---|---|
| Signal Word | Danger |
| Hazard Statements | H314 |
| Precautionary Statements | P280-P305 + P351 + P338-P310 |
| Personal Protective Equipment | Faceshields;full-face respirator (US);Gloves;Goggles;multi-purpose combination respirator cartridge (US);type ABEK (EN14387) respirator filter |
| Hazard Codes | C:Corrosive; |
| Risk Phrases | R34 |
| Safety Phrases | S26-S36/37/39-S45 |
| RIDADR | UN 2735 8/PG 3 |
| WGK Germany | 3 |
| Packaging Group | III |
| Hazard Class | 8 |
| HS Code | 2924199090 |
| Precursor 10 | |
|---|---|
| DownStream 10 | |
| HS Code | 2924199090 |
|---|---|
| Summary | 2924199090. other acyclic amides (including acyclic carbamates) and their derivatives; salts thereof. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:30.0% |
|
Synthesis and antitumor properties of an anthraquinone bisubstituted by the copper chelating peptide Gly-Gly-L-His.
J. Med. Chem. 36 , 2084, (1993) A new molecule 4 [(GGH-DAE)2DHQ] associating the 1,4,5,8-tetrahydroxyanthraquinone ring (DHQ) of the antitumor drug mitoxantrone (2), two diaminoethylene chains (DAE), and the metal-chelating peptide ... |
|
|
A convenient and scalable synthesis of ethyl N-[(2-Boc-amino)ethyl]glycinate and its hydrochloride. Key intermediates for peptide nucleic acid synthesis.
J. Org. Chem. 68 , 1630, (2003) An improved synthesis of ethyl N-[(2-Boc-amino)ethyl]glycinate and its hydrochloride salt is reported. The synthesis is based on the reductive alkylation of Boc-ethylenediamine with ethyl glyoxylate h... |
|
|
Synthesis and evaluation of some nitrobenzenesulfonamides containing nitroisopropyl and (ureidooxy)methyl groups as novel hypoxic cell selective cytotoxic agents.
J. Med. Chem. 34 , 3132, (1991) Basic nitrobenzenesulfonamides containing nitroisopropyl and (ureidooxy)methyl groups were prepared and evaluated as novel hypoxic cell selective cytotoxic agents. In vitro, N-(2-aminoethyl)-N-methyl-... |
| Carbamic acid, N-(2-aminoethyl)-, 1,1-dimethylethyl ester |
| N-tert-Boc-ethylenediamine |
| tert-butyl-(2-aminoethyl)carbamat |
| tert-butyl (2-aminoethyl)carbamate |
| 2-Methyl-2-propanyl (2-aminoethyl)carbamate |
| tert-butyl N-(2-aminoethyl)carbamate |
| MFCD00191871 |
| N-(tert-Butoxycarbonyl)-1,2-diaminoethane |
 CAS#:24424-99-5
CAS#:24424-99-5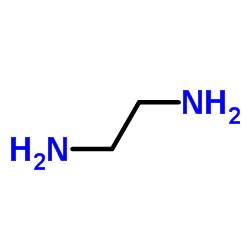 CAS#:107-15-3
CAS#:107-15-3 CAS#:85363-04-8
CAS#:85363-04-8 CAS#:58632-95-4
CAS#:58632-95-4 CAS#:6627-89-0
CAS#:6627-89-0 CAS#:4363-36-4
CAS#:4363-36-4 CAS#:35150-09-5
CAS#:35150-09-5 CAS#:77361-32-1
CAS#:77361-32-1 CAS#:77153-05-0
CAS#:77153-05-0 CAS#:34619-03-9
CAS#:34619-03-9![tert-butyl N-[2-[(2-chloro-3-nitropyridin-4-yl)amino]ethyl]carbamate structure](https://image.chemsrc.com/caspic/454/1061318-65-7.png) CAS#:1061318-65-7
CAS#:1061318-65-7![tert-butyl N-[2-(pyridin-2-ylamino)ethyl]carbamate structure](https://image.chemsrc.com/caspic/190/187339-13-5.png) CAS#:187339-13-5
CAS#:187339-13-5 CAS#:215254-91-4
CAS#:215254-91-4 CAS#:222639-98-7
CAS#:222639-98-7 CAS#:225797-46-6
CAS#:225797-46-6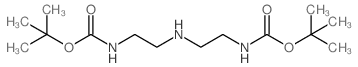 CAS#:117499-16-8
CAS#:117499-16-8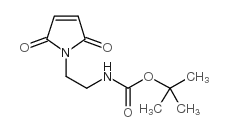 CAS#:134272-63-2
CAS#:134272-63-2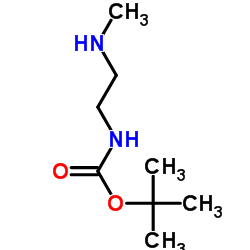 CAS#:122734-32-1
CAS#:122734-32-1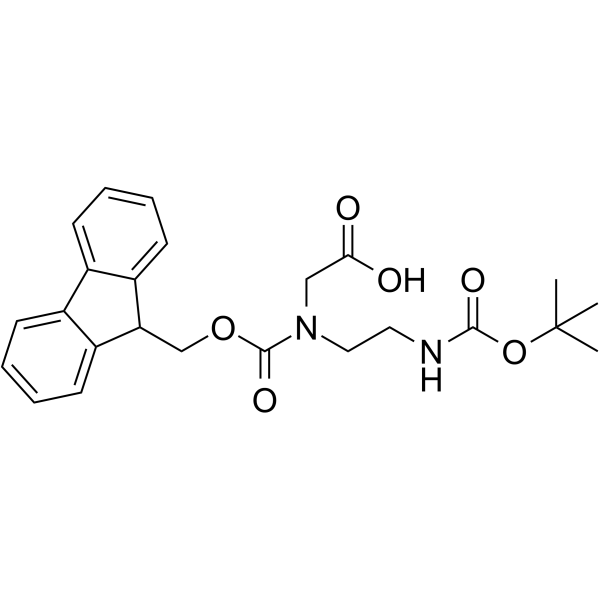 CAS#:141743-15-9
CAS#:141743-15-9
