Oxetane
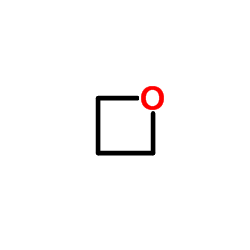
Oxetane structure
|
Common Name | Oxetane | ||
|---|---|---|---|---|
| CAS Number | 503-30-0 | Molecular Weight | 58.079 | |
| Density | 0.9±0.1 g/cm3 | Boiling Point | 49.0±0.0 °C at 760 mmHg | |
| Molecular Formula | C3H6O | Melting Point | -97°C | |
| MSDS | Chinese USA | Flash Point | -32.0±15.3 °C | |
| Symbol |


GHS02, GHS07 |
Signal Word | Danger | |
| Name | oxetane |
|---|---|
| Synonym | More Synonyms |
| Density | 0.9±0.1 g/cm3 |
|---|---|
| Boiling Point | 49.0±0.0 °C at 760 mmHg |
| Melting Point | -97°C |
| Molecular Formula | C3H6O |
| Molecular Weight | 58.079 |
| Flash Point | -32.0±15.3 °C |
| Exact Mass | 58.041866 |
| PSA | 9.23000 |
| LogP | -0.24 |
| Vapour Pressure | 317.0±0.0 mmHg at 25°C |
| Index of Refraction | 1.412 |
| Storage condition | 2-8°C |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
MUTATION DATA
|
| Symbol |


GHS02, GHS07 |
|---|---|
| Signal Word | Danger |
| Hazard Statements | H225-H302-H312-H332 |
| Precautionary Statements | P210-P280 |
| Personal Protective Equipment | Eyeshields;Faceshields;full-face respirator (US);Gloves;multi-purpose combination respirator cartridge (US) |
| Hazard Codes | F:Highlyflammable;Xn:Harmful; |
| Risk Phrases | R11;R20/21/22 |
| Safety Phrases | S9-S16-S26-S29 |
| RIDADR | UN 1993 3/PG 2 |
| WGK Germany | 1 |
| RTECS | RQ6825000 |
| Packaging Group | II |
| Hazard Class | 3 |
| HS Code | 2932999099 |
| Precursor 9 | |
|---|---|
| DownStream 9 | |
| HS Code | 2932999099 |
|---|---|
| Summary | 2932999099. other heterocyclic compounds with oxygen hetero-atom(s) only. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
|
Synthesis of highly substituted oxetanes via [2+2] cycloaddition reactions of allenoates catalyzed by a guanidine Lewis base.
Chem. Commun. (Camb.) 49(28) , 2930-2, (2013) The first synthesis of highly substituted 3-alkyl-oxetan-2-ylidenes from allenoates was developed by using the bicyclic guanidine 1,5,7-triazabicyclo[4.4.0]dec-5-ene (TBD) as an exceptionally active n... |
|
|
A selective intramolecular transacylation of taxoids accompanying with the oxetane ring opening.
Chem. Pharm. Bull. 60(3) , 415-8, (2012) A selective intramolecular transacylation from C-4 to C-5 of taxoids, which occurred simultaneously with the oxetane D-ring opening and was promoted by TiCl4, was presented. The optimal condition was ... |
|
|
Three-component assembly and divergent ring-expansion cascades of functionalized 2-iminooxetanes.
Angew. Chem. Int. Ed. Engl. 49(48) , 9210-4, (2010)
|
| MFCD00005167 |
| Oxacyclobutane |
| Trimethylene oxide/Oxetane |
| 1,3-Epoxypropane |
| Trimethylene Oxide |
| Oxetane |
| Cyclooxabutane |
| EINECS 207-964-3 |
| Trimethylene oxide (8CI) |
 CAS#:287-25-2
CAS#:287-25-2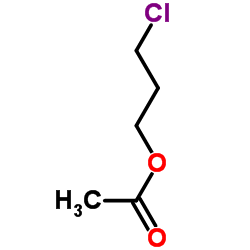 CAS#:628-09-1
CAS#:628-09-1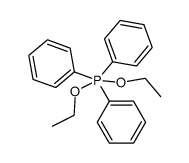 CAS#:86852-11-1
CAS#:86852-11-1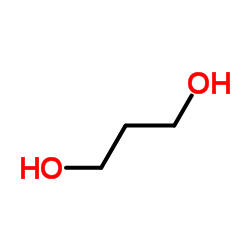 CAS#:504-63-2
CAS#:504-63-2 CAS#:627-18-9
CAS#:627-18-9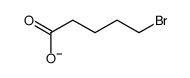 CAS#:592-33-6
CAS#:592-33-6 CAS#:627-30-5
CAS#:627-30-5 CAS#:74-98-6
CAS#:74-98-6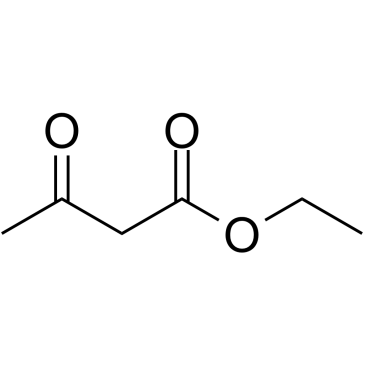 CAS#:141-97-9
CAS#:141-97-9 CAS#:1067-98-7
CAS#:1067-98-7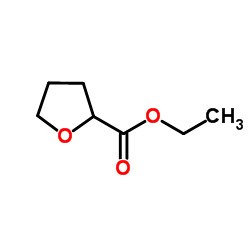 CAS#:16874-34-3
CAS#:16874-34-3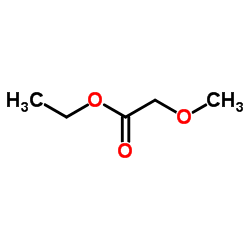 CAS#:3938-96-3
CAS#:3938-96-3 CAS#:17640-26-5
CAS#:17640-26-5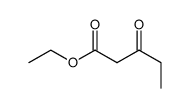 CAS#:15224-07-4
CAS#:15224-07-4 CAS#:96516-90-4
CAS#:96516-90-4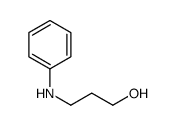 CAS#:31121-11-6
CAS#:31121-11-6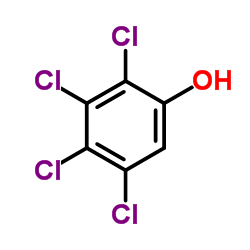 CAS#:4901-51-3
CAS#:4901-51-3 CAS#:133752-76-8
CAS#:133752-76-8