1,4-Dioxaspiro[4.5]decan-8-one
![1,4-Dioxaspiro[4.5]decan-8-one Structure](https://image.chemsrc.com/caspic/201/4746-97-8.png)
1,4-Dioxaspiro[4.5]decan-8-one structure
|
Common Name | 1,4-Dioxaspiro[4.5]decan-8-one | ||
|---|---|---|---|---|
| CAS Number | 4746-97-8 | Molecular Weight | 156.18 | |
| Density | 1.2±0.1 g/cm3 | Boiling Point | 268.3±40.0 °C at 760 mmHg | |
| Molecular Formula | C8H12O3 | Melting Point | 70-73 °C(lit.) | |
| MSDS | Chinese USA | Flash Point | 106.7±13.8 °C | |
Use of 1,4-Dioxaspiro[4.5]decan-8-one1,4-dioxaspiro[4.5]decan-8-one is a biochemical reagent that can be used as a biological material or organic compound for life science related research. |
| Name | 1,4-Dioxaspiro[4.5]decan-8-one |
|---|---|
| Synonym | More Synonyms |
| Description | 1,4-dioxaspiro[4.5]decan-8-one is a biochemical reagent that can be used as a biological material or organic compound for life science related research. |
|---|---|
| Related Catalog | |
| In Vitro | 1,4-Cyclohexanedione 用于制备一系列强效镇痛化合物。1,4-Cyclohexanedione 还用作合成氚标记探针的构建块,用于多巴胺再摄取复合物的放射自显影研究。 |
| Density | 1.2±0.1 g/cm3 |
|---|---|
| Boiling Point | 268.3±40.0 °C at 760 mmHg |
| Melting Point | 70-73 °C(lit.) |
| Molecular Formula | C8H12O3 |
| Molecular Weight | 156.18 |
| Flash Point | 106.7±13.8 °C |
| Exact Mass | 156.078644 |
| PSA | 35.53000 |
| LogP | -0.08 |
| Vapour Pressure | 0.0±0.5 mmHg at 25°C |
| Index of Refraction | 1.489 |
| Personal Protective Equipment | Eyeshields;Gloves;type N95 (US);type P1 (EN143) respirator filter |
|---|---|
| Hazard Codes | Xi:Irritant |
| Risk Phrases | R36/37/38 |
| Safety Phrases | S22-S24/25-S36/37/39-S26 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| HS Code | 2932999099 |
| Precursor 8 | |
|---|---|
| DownStream 10 | |
| HS Code | 2932999099 |
|---|---|
| Summary | 2932999099. other heterocyclic compounds with oxygen hetero-atom(s) only. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
|
A mild method for the preparation of gamma-hydroxy-alpha,beta-acetylenic esters.
Angew. Chem. Int. Ed. Engl. 43 , 2525, (2004)
|
|
|
Aromatization of 1,6,7,7a-tetrahydro-2H-indol-2-ones by a novel process. Preparation of key-intermediate methyl 1-benzyl-5-methoxy-1H-indole-3-acetate and the syntheses of serotonin, melatonin, and bufotenin.
J. Org. Chem. 67 , 2252, (2002) Imine 7 of 1,4-cyclohexanedione mono-ethylene ketal 6 was reacted with maleic anhydride, affording the cyclized adduct 8. Methyl esterification of 8, accompanied by transacetalization, led to the dihy... |
|
|
Aldrichimica Acta 12 , 40, (1979)
|
| 1,4-Cyclohexanedione monoethylene ketal |
| 1,4-Dioxaspiro[4,5]decan-8-one |
| 1,4-cyclohexanedione monoacetal |
| MFCD00010214 |
| 1,4-Cyclohexanedione monoethylene acetal |
| 1,4-Cyclohexanedione monoethyleneacetal |
| 1,4-Dioxaspiro[4.5]decan-8-one |
| 1,4-Cyclohexanedione Monoethyleneketal |
![1,4-dioxaspiro[4.5]deca-6,9-dien-8-one Structure](https://image.chemsrc.com/caspic/325/35357-34-7.png) CAS#:35357-34-7
CAS#:35357-34-7![1,4-Dioxaspiro[4.5]decan-8-ol Structure](https://image.chemsrc.com/caspic/198/22428-87-1.png) CAS#:22428-87-1
CAS#:22428-87-1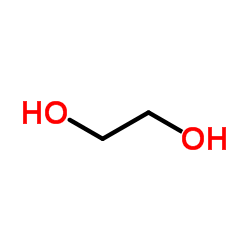 CAS#:107-21-1
CAS#:107-21-1 CAS#:637-88-7
CAS#:637-88-7 CAS#:183-97-1
CAS#:183-97-1![Bis[1,4-dioxaspiro[4.5]decan-8-yliden]hydrazin Structure](https://image.chemsrc.com/caspic/331/64554-48-9.png) CAS#:64554-48-9
CAS#:64554-48-9![ethyl 8-oxo-1,4-dioxaspiro[4.5]decane-7-carboxylate Structure](https://image.chemsrc.com/caspic/086/14160-65-7.png) CAS#:14160-65-7
CAS#:14160-65-7![(4aS,10bR)-6,10b-dimethyl-4a,10b-dihydrodispiro[naphtho[2,1-e][1,2,4]trioxine-3,1'-cyclohexane-4',2''-[1,3]dioxolane] Structure](https://image.chemsrc.com/caspic/191/124293-54-5.png) CAS#:124293-54-5
CAS#:124293-54-5 CAS#:106349-63-7
CAS#:106349-63-7 CAS#:110299-94-0
CAS#:110299-94-0 CAS#:106332-42-7
CAS#:106332-42-7![1,4,9-trioxaspiro[4.6]undecan-8-one structure](https://image.chemsrc.com/caspic/369/110674-74-3.png) CAS#:110674-74-3
CAS#:110674-74-3 CAS#:55145-45-4
CAS#:55145-45-4 CAS#:36716-71-9
CAS#:36716-71-9 CAS#:5432-85-9
CAS#:5432-85-9![8-amino-1,4-dioxaspiro[4.5]decane-8-carboxylic acid structure](https://image.chemsrc.com/caspic/293/54621-18-0.png) CAS#:54621-18-0
CAS#:54621-18-0![8-but-3-enyl-1,4-dioxaspiro[4.5]decan-8-ol structure](https://image.chemsrc.com/caspic/331/142283-61-2.png) CAS#:142283-61-2
CAS#:142283-61-2
