Tetrabutylammonium hexafluorophosphate
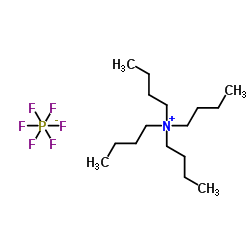
Tetrabutylammonium hexafluorophosphate structure
|
Common Name | Tetrabutylammonium hexafluorophosphate | ||
|---|---|---|---|---|
| CAS Number | 3109-63-5 | Molecular Weight | 387.42800 | |
| Density | N/A | Boiling Point | 242-246 °C | |
| Molecular Formula | C16H36F6NP | Melting Point | 244-246 °C(lit.) | |
| MSDS | Chinese USA | Flash Point | N/A | |
Use of Tetrabutylammonium hexafluorophosphateTetrabutylammonium hexafluorophosphate(V) is a biochemical reagent that can be used as a biological material or organic compound for life science related research. |
| Name | tetrabutylazanium,hexafluorophosphate |
|---|---|
| Synonym | More Synonyms |
| Description | Tetrabutylammonium hexafluorophosphate(V) is a biochemical reagent that can be used as a biological material or organic compound for life science related research. |
|---|---|
| Related Catalog |
| Boiling Point | 242-246 °C |
|---|---|
| Melting Point | 244-246 °C(lit.) |
| Molecular Formula | C16H36F6NP |
| Molecular Weight | 387.42800 |
| Exact Mass | 387.24900 |
| PSA | 13.59000 |
| LogP | 8.38600 |
| InChIKey | BKBKEFQIOUYLBC-UHFFFAOYSA-N |
| SMILES | CCCC[N+](CCCC)(CCCC)CCCC.F[P-](F)(F)(F)(F)F |
| Water Solubility | acetonitrile: 0.1 g/mL, clear, colorless |
| Personal Protective Equipment | dust mask type N95 (US);Eyeshields;Gloves |
|---|---|
| Hazard Codes | Xi:Irritant; |
| Risk Phrases | R36/37/38 |
| Safety Phrases | S26 |
| RIDADR | 1759 |
| WGK Germany | 3 |
|
Astaxanthin diferulate as a bifunctional antioxidant.
Free Radic. Res. 49(1) , 102-11, (2014) Astaxanthin when esterified with ferulic acid is better singlet oxygen quencher with k2 = (1.58 ± 0.1) 10(10) L mol(-1)s(-1) in ethanol at 25°C compared with astaxanthin with k2 = (1.12 ± 0.01) 10(9) ... |
|
|
Cyclometalated iridium(III) bipyridyl-phenylenediamine complexes with multicolor phosphorescence: synthesis, electrochemistry, photophysics, and intracellular nitric oxide sensing.
ChemMedChem 9(6) , 1316-29, (2014) We present a new class of phosphorescent cyclometalated iridium(III) bipyridyl-phenylenediamine complexes [Ir(N^C)2 (bpy-DA)](PF6 ) (bpy-DA=4-(N-(2-amino-5-methoxyphenyl)aminomethyl)-4'-methyl-2,2'-bi... |
|
|
A quantitative assay for reductive metabolism of a pesticide in fish using electrochemistry coupled with liquid chromatography tandem mass spectrometry.
Environ. Sci. Technol. 49(7) , 4450-7, (2015) This is the first study to use electrochemistry to generate a nitro reduction metabolite as a standard for a liquid chromatography-mass spectrometry-based quantitative assay. This approach is further ... |
| 1-Butanaminium, N,N,N-tributyl-, hexafluorophosphate(1-) |
| EINECS 221-472-6 |
| Tetrabutylammonium hexafluorophosphate |
| MFCD00011748 |
| N,N,N-Tributyl-1-butanaminium hexafluorophosphate(1-) |
| 4K4&4&4 &&PF6 salt |
| N,N,N-Tributyl-1-butanaminium hexafluorophosphate |
| N,N,N-Tributylbutan-1-aminium hexafluorophosphate |
| Tetra-n-butylammonium hexafluorophosphate |

