GERANYLGERANIOL

GERANYLGERANIOL structure
|
Common Name | GERANYLGERANIOL | ||
|---|---|---|---|---|
| CAS Number | 24034-73-9 | Molecular Weight | 290.48300 | |
| Density | 0.88 g/cm3 | Boiling Point | 397.1ºC at 760 mmHg | |
| Molecular Formula | C20H34O | Melting Point | N/A | |
| MSDS | Chinese USA | Flash Point | 128.3ºC | |
| Symbol |

GHS07 |
Signal Word | Warning | |
Use of GERANYLGERANIOLGeranylgeraniol is an orally acitve vitamin K2 sub-type, an intermediate of the mevalonate pathway. Geranylgeraniol targets NF-kB signaling pathway and could alleviate LPS-induced microglial inflammation in animal model[1][2][3][4]. |
| Name | (E,E,E)-geranylgeraniol |
|---|---|
| Synonym | More Synonyms |
| Description | Geranylgeraniol is an orally acitve vitamin K2 sub-type, an intermediate of the mevalonate pathway. Geranylgeraniol targets NF-kB signaling pathway and could alleviate LPS-induced microglial inflammation in animal model[1][2][3][4]. |
|---|---|
| Related Catalog | |
| Target |
NF-kB[1] |
| In Vitro | Geranylgeraniol (0-10 μM; 24 h) dose-dependently suppresses the LPS-induced increase in the mRNA levels of Il-1β, Tnf-α, Il-6, and Cox-2[1]. Geranylgeraniol (10 μM; 24 h) inhibits the phosphorylation of TAK1, IKKα/β, and NF-κB p65 proteins as well as NF-κB nuclear translocation induced by LPS while maintaining IκBα expression[1]. Geranylgeraniol, (50 μM; 24 h) eliminates cell damage caused by Simvastatin (HY-17502) (10 µM) and Mevalonat (10 mM), and reduces the inflammatory marker and the damage of the mitochondria, maintaining its shape and component[2]. Western Blot Analysis[1] Cell Line: MG6 cell Concentration: 0, 1, 10 μM Incubation Time: 24 hours Result: Suppressed by TAK1, IKKα/β, and NF-κB p65 proteins level at 10 μM. RT-PCR[1] Cell Line: MG6 cell Concentration: 10 μM Incubation Time: 0, 6, 12, 24 hours Result: Significantly inhibited pro-inflammatory cytokine Il-1β, Tnf-α, Il-6, and Cox-2 mRNA level. |
| In Vivo | Geranylgeraniol (725 mg/kg/d; p.o.; 90 d) is not toxicologically significant with a dose below 725 mg/kg/d in rats[3]. Geranylgeraniol (483 mg/kg/d; p.o.; 10 d) suppresses lipopolysaccharide-induced inflammation via inhibition of nuclear factor-κB activation in rats[4]. Animal Model: Han Wistar rats (169-192 g for male; 116-152 g for female)[3] Dosage: 0, 725, 1450, and 2900 mg/kg Administration: Oral gavage; once daily; 90 days Result: Showed the lowest observed adverse effect level (LOAEL) for local effects and the no observed adverse effect level (NOAEL) for systemic effects as 725 mg/kg/d. Reduced body weights by 12.9 and 21.6% in the intermediate- and high-dose group males, respectively, compared to controls. Animal Model: Wistar rats (male, 8-week-old, 130-150 g)[4] Dosage: 0, 48.3, 483, 4830 mg/kg Administration: Oral gavage; once daily; 10 days; with or not LPS challenge (i.p.; 0.5 mg/kg) Result: Suppressed LPS-induced inflammatory cytokines and mRNA expression of LPS-induced inflammatory genes in liver with doses of 483 mg/kg and 4830 mg/kg. Suppressed protein levels of IRAK1, TRAF6, and TAK1, originating from transcriptional down-regulation with doses of 483 mg/kg and 4830 mg/kg. |
| References |
| Density | 0.88 g/cm3 |
|---|---|
| Boiling Point | 397.1ºC at 760 mmHg |
| Molecular Formula | C20H34O |
| Molecular Weight | 290.48300 |
| Flash Point | 128.3ºC |
| Exact Mass | 290.26100 |
| PSA | 20.23000 |
| LogP | 6.12440 |
| Vapour Pressure | 0mmHg at 25°C |
| Index of Refraction | 1.492 |
| Storage condition | -20°C |
| Symbol |

GHS07 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H315-H319-H335 |
| Precautionary Statements | P261-P305 + P351 + P338 |
| Personal Protective Equipment | Eyeshields;full-face respirator (US);Gloves;multi-purpose combination respirator cartridge (US);type ABEK (EN14387) respirator filter |
| Hazard Codes | Xi: Irritant; |
| Risk Phrases | R36/37/38 |
| Safety Phrases | 26-36 |
| RIDADR | NONH for all modes of transport |
| HS Code | 2905290000 |
| Precursor 4 | |
|---|---|
| DownStream 10 | |
| HS Code | 2905290000 |
|---|---|
| Summary | 2905290000 unsaturated monohydric alcohols。Supervision conditions:None。VAT:17.0%。Tax rebate rate:9.0%。MFN tariff:5.5%。General tariff:30.0% |
|
Polyunsaturated Branched-Chain Fatty Acid Geranylgeranoic Acid Induces Unfolded Protein Response in Human Hepatoma Cells.
PLoS ONE 10 , e0132761, (2015) The acyclic diterpenoid acid geranylgeranoic acid (GGA) has been reported to induce autophagic cell death in several human hepatoma-derived cell lines; however, the molecular mechanism for this remain... |
|
|
PBK/TOPK mediates geranylgeranylation signaling for breast cancer cell proliferation.
Cancer Cell Int. 15 , 27, (2015) PDZ binding-kinase (PBK) (also named T-lymphokine-activated killer cell-originated protein kinase (TOPK)), a serine/threonine kinase, is tightly controlled in normal tissues but elevated in many tumor... |
|
|
Novel High Content Screen Detects Compounds That Promote Neurite Regeneration from Cochlear Spiral Ganglion Neurons.
Sci. Rep. 5 , 15960, (2015) The bipolar spiral ganglion neurons (SGN) carry sound information from cochlear hair cells to the brain. After noise, antibiotic or toxic insult to the cochlea, damage to SGN and/or hair cells causes ... |
| MFCD00129083 |
| Geranylgeraniol |
| (2E,6E,10E)-3,7,11,15-tetramethylhexadeca-2,6,10,14-tetraen-1-ol |
| all trans-3,7,11-15-Tetramethyl-2,6,10,14-hexadecatetraen-1-ol |
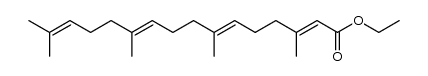 CAS#:24035-35-6
CAS#:24035-35-6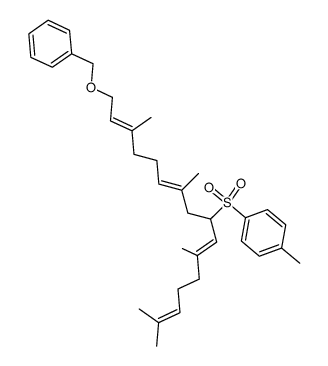 CAS#:68690-51-7
CAS#:68690-51-7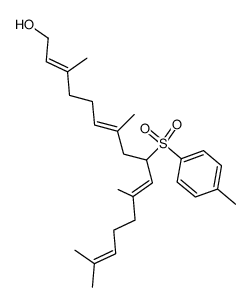 CAS#:71816-56-3
CAS#:71816-56-3 CAS#:24163-93-7
CAS#:24163-93-7![[3-methyl-3-(4,8,12-trimethyltridecyl)oxiran-2-yl]methanol structure](https://image.chemsrc.com/caspic/455/107438-44-8.png) CAS#:107438-44-8
CAS#:107438-44-8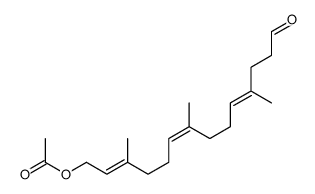 CAS#:142673-23-2
CAS#:142673-23-2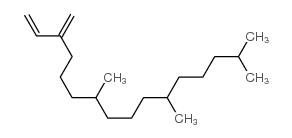 CAS#:504-96-1
CAS#:504-96-1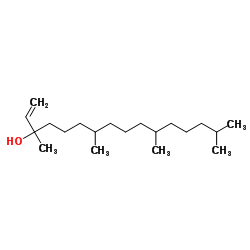 CAS#:505-32-8
CAS#:505-32-8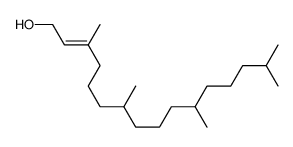 CAS#:7541-49-3
CAS#:7541-49-3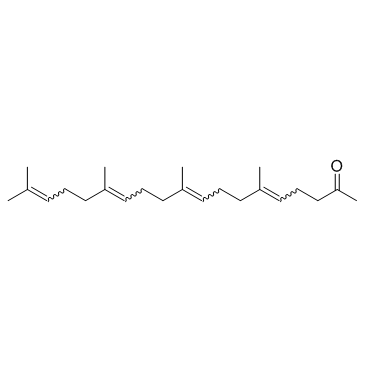 CAS#:6809-52-5
CAS#:6809-52-5 CAS#:13190-97-1
CAS#:13190-97-1 CAS#:59-02-9
CAS#:59-02-9 CAS#:2140-82-1
CAS#:2140-82-1 CAS#:6699-20-3
CAS#:6699-20-3
