Thiabendazole
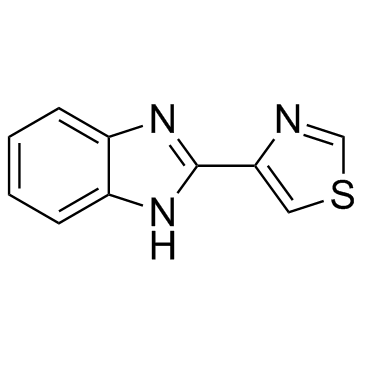
Thiabendazole structure
|
Common Name | Thiabendazole | ||
|---|---|---|---|---|
| CAS Number | 148-79-8 | Molecular Weight | 201.248 | |
| Density | 1.4±0.1 g/cm3 | Boiling Point | 446.0±37.0 °C at 760 mmHg | |
| Molecular Formula | C10H7N3S | Melting Point | 298-301ºC | |
| MSDS | Chinese USA | Flash Point | 226.2±16.9 °C | |
| Symbol |

GHS09 |
Signal Word | Warning | |
Use of ThiabendazoleThiabendazole inhibites the mitochondrial helminth-specific enzyme, fumarate reductase, with anthelminthic property. Target: Fumarate ReductaseTiabendazole serves to block angiogenesis in both frog embryos and human cells. It has also been shown to serve as a vascular disrupting agent to reduce newly established blood vessels. Tiabendazole has been shown to effectively do this in certain cancer cells. Thiabendazole works by inhibition of the mitochondrial, helminth-specific enzyme, fumarate reductase, with possible interaction with endogenous quinone [1].Thiabendazole inhibited B16F10 proliferation in vitro in a dose- and time-dependent manner with an IC50 of 532.4 +/- 32.6, 322.9 +/- 28.9, 238.5 +/- 19.8 microM at 24, 48, and 72 h, respectively. Moreover, thiabendazole inhibited the angiogenesis and the migration of B16F10 cells in vitro. Furthermore, thiabendazole restrained transcription and translation of the VEGF gene in B16F10 in vitro, and the apoptotic percentage of B16F10 cells was increased after exposure to thiabendazole [2]. |
| Name | thiabendazole |
|---|---|
| Synonym | More Synonyms |
| Description | Thiabendazole inhibites the mitochondrial helminth-specific enzyme, fumarate reductase, with anthelminthic property. Target: Fumarate ReductaseTiabendazole serves to block angiogenesis in both frog embryos and human cells. It has also been shown to serve as a vascular disrupting agent to reduce newly established blood vessels. Tiabendazole has been shown to effectively do this in certain cancer cells. Thiabendazole works by inhibition of the mitochondrial, helminth-specific enzyme, fumarate reductase, with possible interaction with endogenous quinone [1].Thiabendazole inhibited B16F10 proliferation in vitro in a dose- and time-dependent manner with an IC50 of 532.4 +/- 32.6, 322.9 +/- 28.9, 238.5 +/- 19.8 microM at 24, 48, and 72 h, respectively. Moreover, thiabendazole inhibited the angiogenesis and the migration of B16F10 cells in vitro. Furthermore, thiabendazole restrained transcription and translation of the VEGF gene in B16F10 in vitro, and the apoptotic percentage of B16F10 cells was increased after exposure to thiabendazole [2]. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.4±0.1 g/cm3 |
|---|---|
| Boiling Point | 446.0±37.0 °C at 760 mmHg |
| Melting Point | 298-301ºC |
| Molecular Formula | C10H7N3S |
| Molecular Weight | 201.248 |
| Flash Point | 226.2±16.9 °C |
| Exact Mass | 201.036072 |
| PSA | 69.81000 |
| LogP | 2.47 |
| Vapour Pressure | 0.0±1.1 mmHg at 25°C |
| Index of Refraction | 1.740 |
| Water Solubility | 0.005 g/100 mL |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
MUTATION DATA
|
| Symbol |

GHS09 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H410 |
| Precautionary Statements | P273-P501 |
| Personal Protective Equipment | Eyeshields;Gloves |
| Hazard Codes | N:Dangerousfortheenvironment; |
| Risk Phrases | R50/53 |
| Safety Phrases | S60-S61 |
| RIDADR | UN 3077 |
| WGK Germany | 2 |
| RTECS | DE0700000 |
| Packaging Group | III |
| Hazard Class | 9 |
| HS Code | 2934100015 |
|
~86%
Detail
|
| Literature: E. I. Du Pont de Nemours and Company Patent: US5310923 A1, 1994 ; |
|
~91%
Detail
|
| Literature: E. I. Du Pont de Nemours and Company Patent: US5310924 A1, 1994 ; |
|
~91% 
Thiabendazole CAS#:148-79-8 |
| Literature: E. I. Du Pont de Nemours and Company Patent: US5310924 A1, 1994 ; |
|
~% 
Thiabendazole CAS#:148-79-8 |
| Literature: Journal of the American Chemical Society, , vol. 132, p. 1230 - 1231 |
|
Detail
|
| Literature: E. I. Du Pont de Nemours and Company Patent: US5310923 A1, 1994 ; |
|
~% 
Thiabendazole CAS#:148-79-8 |
| Literature: US2009/142265 A1, ; US 20090142265 A1 |
|
~% 
Thiabendazole CAS#:148-79-8 |
| Literature: Bulletin of the Korean Chemical Society, , vol. 34, # 8 p. 2305 - 2310 |
| Precursor 10 | |
|---|---|
| DownStream 10 | |
| HS Code | 2934100015 |
|---|---|
| Summary | 2934100015 2,4-dimethyl-n-phenylthiazole-5-carboxamide。supervision conditions:s(import or export registration certificate for pesticides)。VAT:17.0%。tax rebate rate:9.0%。MFN tarrif:6.5%。general tariff:20.0% |
|
Cheminformatics analysis of assertions mined from literature that describe drug-induced liver injury in different species.
Chem. Res. Toxicol. 23 , 171-83, (2010) Drug-induced liver injury is one of the main causes of drug attrition. The ability to predict the liver effects of drug candidates from their chemical structures is critical to help guide experimental... |
|
|
Translating clinical findings into knowledge in drug safety evaluation--drug induced liver injury prediction system (DILIps).
J. Sci. Ind. Res. 65(10) , 808, (2006) Drug-induced liver injury (DILI) is a significant concern in drug development due to the poor concordance between preclinical and clinical findings of liver toxicity. We hypothesized that the DILI typ... |
|
|
Pen-on-paper approach toward the design of universal surface enhanced Raman scattering substrates.
Small 10(15) , 3065-71, (2014) The translation of a technology from the laboratory into the real world should meet the demand of economic viability and operational simplicity. Inspired by recent advances in conductive ink pens for ... |
| Arbotect |
| Tresaderm |
| 2-(thiazol-4-yl)benzimidazole |
| Mintezol |
| 2-(4-Thiazolyl)benzimidazole,Thiabendazole |
| 2-(4-thiazolyl)-1H-benzimidazole |
| TBZ |
| Benzimidazole, 2-(4-thiazolyl)- |
| 2-(4-Thiazolyl)benzimidazole |
| 4-(1H-Benzo[d]imidazol-2-yl)thiazole |
| 2-(4-Thiazoly)benzimidazole |
| 1H-Benzimidazole, 2-(4-thiazolyl)- |
| EINECS 205-725-8 |
| Tiabendazole |
| 2-(1,3-Thiazol-4-yl)-1H-benzimidazole |
| 2-(1,3-thiazol-4-yl)benzimidazole |
| MFCD00005587 |
| Thiabendazole |
| 2-(1,3-thiazol-4-yl)-1H-1,3-benzimidazole |
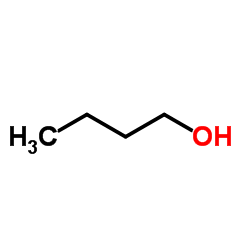
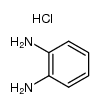
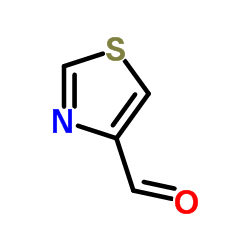


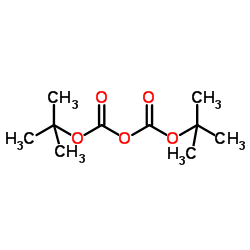
 CAS#:51-17-2
CAS#:51-17-2 CAS#:5805-52-7
CAS#:5805-52-7 CAS#:939-70-8
CAS#:939-70-8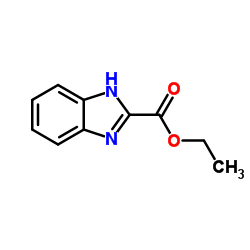 CAS#:1865-09-4
CAS#:1865-09-4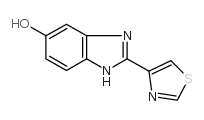 CAS#:948-71-0
CAS#:948-71-0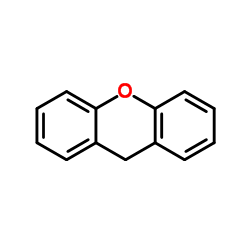 CAS#:92-83-1
CAS#:92-83-1![4-(5-nitro-1H-benzo[d]imidazol-2-yl)thiazole structure](https://image.chemsrc.com/caspic/338/3575-05-1.png) CAS#:3575-05-1
CAS#:3575-05-1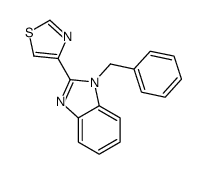 CAS#:33705-43-0
CAS#:33705-43-0 CAS#:32594-70-0
CAS#:32594-70-0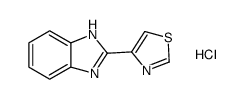 CAS#:945-65-3
CAS#:945-65-3
