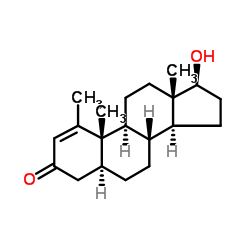
Mesterolone
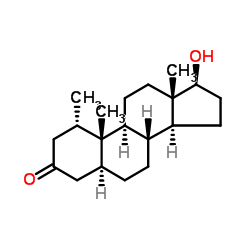
Mesterolone structure
|
Common Name | Mesterolone | ||
|---|---|---|---|---|
| CAS Number | 1424-00-6 | Molecular Weight | 304.467 | |
| Density | 1.1±0.1 g/cm3 | Boiling Point | 420.3±45.0 °C at 760 mmHg | |
| Molecular Formula | C20H32O2 | Melting Point | 208 °C | |
| MSDS | Chinese USA | Flash Point | 179.4±21.3 °C | |
| Symbol |

GHS07 |
Signal Word | Warning | |
| Name | Mesterolone |
|---|---|
| Synonym | More Synonyms |
| Density | 1.1±0.1 g/cm3 |
|---|---|
| Boiling Point | 420.3±45.0 °C at 760 mmHg |
| Melting Point | 208 °C |
| Molecular Formula | C20H32O2 |
| Molecular Weight | 304.467 |
| Flash Point | 179.4±21.3 °C |
| Exact Mass | 304.240234 |
| PSA | 37.30000 |
| LogP | 4.24 |
| Vapour Pressure | 0.0±2.2 mmHg at 25°C |
| Index of Refraction | 1.525 |
| Storage condition | 2-8°C |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Symbol |

GHS07 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H302-H312-H332 |
| Precautionary Statements | P280 |
| Personal Protective Equipment | dust mask type N95 (US);Eyeshields;Gloves |
| Hazard Codes | Xn:Harmful |
| Risk Phrases | R20/21/22 |
| Safety Phrases | S22-S24/25-S36/37/39 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| RTECS | BV8063400 |
| HS Code | 2942000000 |
|
~97% 
Mesterolone CAS#:1424-00-6 |
| Literature: ORTMANN, Ingo; Haeselhoff, Claus Christian Patent: US2008/300428 A1, 2008 ; Location in patent: Page/Page column 1 ; |
|
~% 
Mesterolone CAS#:1424-00-6 |
| Literature: Liebigs Annalen der Chemie, , # 11 p. 1996 - 1999 |
|
~% 
Mesterolone CAS#:1424-00-6 |
| Literature: Liebigs Annalen der Chemie, , # 11 p. 1996 - 1999 |
|
~% 
Mesterolone CAS#:1424-00-6 |
| Literature: Collection of Czechoslovak Chemical Communications, , vol. 30, p. 3468 - 3472 |
|
~% 
Mesterolone CAS#:1424-00-6 |
| Literature: Collection of Czechoslovak Chemical Communications, , vol. 29, p. 3089 - 3095 |
|
~% 
Mesterolone CAS#:1424-00-6 |
| Literature: Collection of Czechoslovak Chemical Communications, , vol. 29, p. 3089 - 3095 |
| HS Code | 2942000000 |
|---|
|
An updated steroid benchmark set and its application in the discovery of novel nanomolar ligands of sex hormone-binding globulin.
J. Med. Chem. 51 , 2047-56, (2008) A benchmark data set of steroids with known affinity for sex hormone-binding globulin (SHBG) has been widely used to validate popular molecular field-based QSAR techniques. We have expanded the data s... |
|
|
Screening for anabolic steroids in urine of forensic cases using fully automated solid phase extraction and LC-MS-MS.
J. Anal. Toxicol. 38(9) , 637-44, (2014) A screening method for 18 frequently measured exogenous anabolic steroids and the testosterone/epitestosterone (T/E) ratio in forensic cases has been developed and validated. The method involves a ful... |
|
|
Osteoporosis in male hypogonadism: responses to androgen substitution differ among men with primary and secondary hypogonadism.
Horm. Res. 60(1) , 21-8, (2003) No randomized study exists comparing the effects of different modes of androgen substitution on bone mineral density (BMD).We performed a prospective, randomized, trial assigning 53 hypogonadal men to... |
| 17β-Hydroxy-1α-methyl-5α-androstan-3-one |
| EINECS 215-836-3 |
| 4,5a-Dihydro-1a-methyltestosterone |
| 1a-Methyl-5a-dihydrotestosterone |
| (1a,5a,17b)-17-Hydroxy-1-methylandrostan-3-one |
| Proviron |
| 1a-Methyl-5a-androstan-17b-ol-3-one |
| 17b-Hydroxy-1a-methyl-5a-androstan-3-one |
| 5α-Androstan-3-one, 17β-hydroxy-1α-methyl- |
| Mesterolone |
| (1S,5S,8R,9S,10S,13S,14S,17S)-17-hydroxy-1,10,13-trimethyl-1,2,4,5,6,7,8,9,11,12,14,15,16,17-tetradecahydrocyclopenta[a]phenanthren-3-one |
| 1α-Methyl-5α-dihydrotestosterone |
| MFCD00133082 |
| (1α,5α,17β)-17-Hydroxy-1-methylandrostan-3-one |
| Androstan-3-one, 17-hydroxy-1-methyl-, (1α,5α,17β)- |