Nitidine chloride
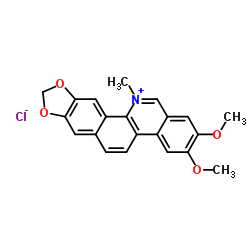
Nitidine chloride structure
|
Common Name | Nitidine chloride | ||
|---|---|---|---|---|
| CAS Number | 13063-04-2 | Molecular Weight | 383.825 | |
| Density | 1.35g/cm3 | Boiling Point | 619ºC at 760 mmHg | |
| Molecular Formula | C21H18ClNO4 | Melting Point | 281-282ºC | |
| MSDS | Chinese USA | Flash Point | 189.4ºC | |
Use of Nitidine chlorideNitidine chloride, a potential anti-malarial lead compound derived from Zanthoxylum nitidum (Roxb) DC, exerts potent anticancer activity through diverse pathways, including inducing apoptosis, inhibiting STAT3 signaling cascade, DNA topoisomerase 1 and 2A, ERK and c-Src/FAK associated signaling pathway. Nitidine chloride inhibits LPS-induced inflammatory cytokines production via MAPK and NF-kB pathway[1][2][3][4][5][6]. |
| Name | 2,3-Dimethoxy-12-methyl-[1,3]dioxolo[4',5':4,5]benzo[1,2-c]phenanthridin-12-ium chloride |
|---|---|
| Synonym | More Synonyms |
| Description | Nitidine chloride, a potential anti-malarial lead compound derived from Zanthoxylum nitidum (Roxb) DC, exerts potent anticancer activity through diverse pathways, including inducing apoptosis, inhibiting STAT3 signaling cascade, DNA topoisomerase 1 and 2A, ERK and c-Src/FAK associated signaling pathway. Nitidine chloride inhibits LPS-induced inflammatory cytokines production via MAPK and NF-kB pathway[1][2][3][4][5][6]. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.35g/cm3 |
|---|---|
| Boiling Point | 619ºC at 760 mmHg |
| Melting Point | 281-282ºC |
| Molecular Formula | C21H18ClNO4 |
| Molecular Weight | 383.825 |
| Flash Point | 189.4ºC |
| Exact Mass | 383.092438 |
| PSA | 40.80000 |
| LogP | 3.71660 |
| Appearance of Characters | white to beige |
| Vapour Pressure | 3.01E-15mmHg at 25°C |
| Index of Refraction | 1.696 |
| InChIKey | QLDAACVSUMUMOR-UHFFFAOYSA-M |
| SMILES | COc1cc2c[n+](C)c3c4cc5c(cc4ccc3c2cc1OC)OCO5.[Cl-] |
| Storage condition | ?20°C |
| Water Solubility | DMSO: soluble1mg/mL, clear (warmed) |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
MUTATION DATA
|
| Safety Phrases | 24/25 |
|---|---|
| RIDADR | NONH for all modes of transport |
| RTECS | DF4935500 |
| HS Code | 2933990090 |
| HS Code | 2933990090 |
|---|---|
| Summary | 2933990090. heterocyclic compounds with nitrogen hetero-atom(s) only. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
|
Nitidine chloride inhibits hepatocellular carcinoma cell growth in vivo through the suppression of the JAK1/STAT3 signaling pathway.
Int. J. Mol. Med. 32(1) , 79-84, (2013) Signal transducer and activator of transcription 3 (STAT3) is persistently activated in cancer cells and contributes to malignant progression in various types of cancer. The Janus-activated kinase (JA... |
|
|
Alkaloids, amides and antispasmodic activity of Zanthoxylum hyemale.
Planta Med. 68(6) , 534-8, (2002) Two quinoline alkaloids, (-)-R-geilbalansine (1) and hyemaline (2), as well as aromatic amide, N-[2-(3,4-dimethoxyphenyl)-2-methoxyethyl)-2-methoxyethyl]benzamide (O-methylbalsamide) (3), were isolate... |
|
|
The role of the iminium bond in the inhibition of reverse transcriptase by quaternary benzophenanthridines.
J. Pharm. Pharmacol. 50(11) , 1307-15, (1998) The quaternary benzo[c]phenanthridines fagaronine, nitidine and O-methylfagaronine have been reviewed as potential antitumour and antiviral agents. Their mode of action has not been established, but t... |
| 2,3-Dimethoxy-12-methyl-[1,3]benzodioxolo[5,6-c]phenanthridinium chloride |
| [1,3]Benzodioxolo[5,6-c]phenanthridinium, 2,3-dimethoxy-12-methyl-, chloride (1:1) |
| 2,3-Dimethoxy-12-methyl[1,3]benzodioxolo[5,6-c]phenanthridin-12-ium chloride |
| Nitidine chloride |
| 2,3-dimethoxy-12-methyl-[1,3]benzodioxolo[5,6-c]phenanthridin-12-ium,chloride |
 CAS#:548-31-2
CAS#:548-31-2 CAS#:105594-89-6
CAS#:105594-89-6![[1,3]Benzodioxolo[5,6-c]phenanthridine,12,13-dihydro-2,3-dimethoxy-12-methyl- Structure](https://image.chemsrc.com/caspic/379/13063-06-4.png) CAS#:13063-06-4
CAS#:13063-06-4![7,8-Dihydronaphtho[2,3-d][1,3]dioxol-5(6H)-one Structure](https://image.chemsrc.com/caspic/113/41303-45-1.png) CAS#:41303-45-1
CAS#:41303-45-1![6,6-dibromo-7,8-dihydronaphtho[2,3-d][1,3]dioxol-5(6H)-one Structure](https://image.chemsrc.com/caspic/278/180411-09-0.png) CAS#:180411-09-0
CAS#:180411-09-0 CAS#:180411-13-6
CAS#:180411-13-6![2,3-dimethoxy-[1,3]benzodioxolo[5,6-c]phenanthridine structure](https://image.chemsrc.com/caspic/379/18034-03-2.png) CAS#:18034-03-2
CAS#:18034-03-2
