Ammonium sulfide
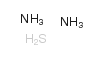
Ammonium sulfide structure
|
Common Name | Ammonium sulfide | ||
|---|---|---|---|---|
| CAS Number | 12135-76-1 | Molecular Weight | 68.14190 | |
| Density | 1 g/mL at 25 °C | Boiling Point | 40 °C | |
| Molecular Formula | H8N2S | Melting Point | -18ºC | |
| MSDS | Chinese USA | Flash Point | 90 °F | |
| Symbol |



GHS02, GHS05, GHS09 |
Signal Word | Danger | |
| Name | Ammonium sulfide |
|---|---|
| Synonym | More Synonyms |
| Density | 1 g/mL at 25 °C |
|---|---|
| Boiling Point | 40 °C |
| Melting Point | -18ºC |
| Molecular Formula | H8N2S |
| Molecular Weight | 68.14190 |
| Flash Point | 90 °F |
| Exact Mass | 68.04080 |
| PSA | 31.78000 |
| LogP | 0.76060 |
| Index of Refraction | n20/D 1.467 |
| Stability | Stable. Pure material is highly flammable - note wide explosion limits. Incompatible with strong oxidizing agents, strong acids, copper, brass, bronze, strong bases, zinc, aluminium. |
| Symbol |



GHS02, GHS05, GHS09 |
|---|---|
| Signal Word | Danger |
| Hazard Statements | H226-H314-H400 |
| Supplemental HS | Contact with acids liberates toxic gas. |
| Precautionary Statements | P210-P260-P280-P303 + P361 + P353-P305 + P351 + P338-P370 + P378 |
| Personal Protective Equipment | Faceshields;full-face respirator (US);Gloves;Goggles;multi-purpose combination respirator cartridge (US);type ABEK (EN14387) respirator filter |
| Hazard Codes | F:Flammable;C:Corrosive;N:Dangerousfortheenvironment; |
| Risk Phrases | R11;R31;R34;R50 |
| Safety Phrases | S26-S36/37/39-S45-S61 |
| RIDADR | UN 2683 8/PG 2 |
| WGK Germany | - |
| Packaging Group | II |
| Hazard Class | 8 |
| HS Code | 2830909000 |
|
~% 
Ammonium sulfide CAS#:12135-76-1 |
| Literature: Journal of the American Chemical Society, , vol. 48, p. 2319 - 2327 |
|
~% 
Ammonium sulfide CAS#:12135-76-1 |
| Literature: Journal fuer Praktische Chemie (Leipzig), , vol. 7, p. 475 Journal fuer Praktische Chemie (Leipzig), , vol. 7, p. 476 |
|
~% 
Ammonium sulfide CAS#:12135-76-1 |
| Literature: Journal of the Chemical Society, , vol. 29, p. 648 N: MVol.2, 5, page 479 - 485 |
| Precursor 4 | |
|---|---|
| DownStream 7 | |
| HS Code | 2830909000 |
|---|
|
Characterization of volatile compounds from the reaction of 3-hydroxy-2-butanone and ammonium sulfide model system.
J. Agric. Food Chem. 47(1) , 245-8, (1999) The reactions between 3-hydroxy-2-butanone and ammoniun sulfide at 25, 50, 75, 100, 125, and 150 degrees C were studied. Four well-known flavor compounds, 2,4,5-trimethyloxazole, 2,4, 5-trimethyl-3-ox... |
|
|
A cataluminescence gas sensor for ammonium sulfide based on Fe(3)O(4)-carbon nanotubes composite.
Luminescence 25(4) , 294-9, (2010) In the present work, Fe(3)O(4)-carbon nanotubes (CNTs) composite was explored as a sensing material candidate for ammonium sulfide. Intense chemiluminescence emission can be observed during the cataly... |
|
|
Visualization of iron in cultured macrophages: a cytochemical light and electron microscopic study using autometallography.
Free Radic. Biol. Med. 15(1) , 1-11, (1993) The objective of this study was to develop a sensitive cytochemical method for the visualization of iron, both at light microscopical (LM) and at electron microscopical (EM) levels, in glutaraldehyde-... |
| Ammonium bisulfide |
| Ammoniumsulfid |
| AMMONIUM SULPHIDE |
| Ammonium Sulfide |
| trueammoniumsulfide |
| EINECS 235-223-4 |
| FEMA 2053 |
| NH4HS |
| diammoniumsulfide |
| ammonium sulfude |
| ammonium monosulfide |
| MFCD00010892 |

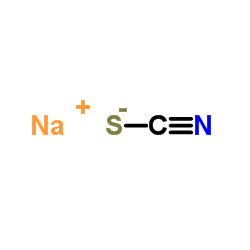 CAS#:540-72-7
CAS#:540-72-7 CAS#:497-19-8
CAS#:497-19-8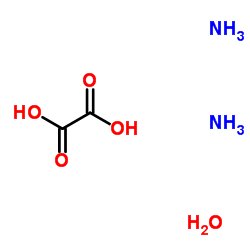 CAS#:6009-70-7
CAS#:6009-70-7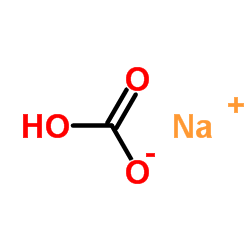 CAS#:144-55-8
CAS#:144-55-8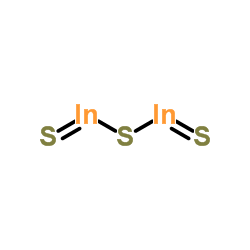 CAS#:12030-24-9
CAS#:12030-24-9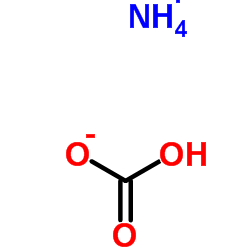 CAS#:1066-33-7
CAS#:1066-33-7