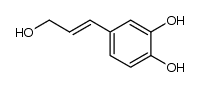
4-allylcatechol
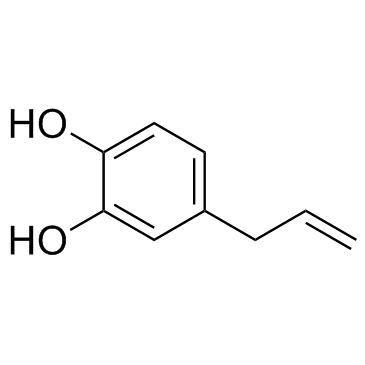
4-allylcatechol structure
|
Common Name | 4-allylcatechol | ||
|---|---|---|---|---|
| CAS Number | 1126-61-0 | Molecular Weight | 150.174 | |
| Density | 1.1±0.1 g/cm3 | Boiling Point | 289.2±25.0 °C at 760 mmHg | |
| Molecular Formula | C9H10O2 | Melting Point | 42.0 to 46.0 °C | |
| MSDS | Chinese USA | Flash Point | 141.7±17.8 °C | |
Use of 4-allylcatechol4-Allylcatechol (4-Allylpyrocatechol, Hydroxychavicol) is an intermediate to synthetic safrole. |
| Name | 4-Allylpyrocatechol |
|---|---|
| Synonym | More Synonyms |
| Description | 4-Allylcatechol (4-Allylpyrocatechol, Hydroxychavicol) is an intermediate to synthetic safrole. |
|---|---|
| Related Catalog |
| Density | 1.1±0.1 g/cm3 |
|---|---|
| Boiling Point | 289.2±25.0 °C at 760 mmHg |
| Melting Point | 42.0 to 46.0 °C |
| Molecular Formula | C9H10O2 |
| Molecular Weight | 150.174 |
| Flash Point | 141.7±17.8 °C |
| Exact Mass | 150.068085 |
| PSA | 40.46000 |
| LogP | 1.90 |
| Vapour Pressure | 0.0±0.6 mmHg at 25°C |
| Index of Refraction | 1.587 |
| Storage condition | 2-8°C |
| RIDADR | NONH for all modes of transport |
|---|---|
| RTECS | CZ9029440 |
| HS Code | 2907299090 |
| Precursor 10 | |
|---|---|
| DownStream 7 | |
| HS Code | 2907299090 |
|---|---|
| Summary | 2907299090 polyphenols; phenol-alcohols。supervision conditions:AB(certificate of inspection for goods inward,certificate of inspection for goods outward)。VAT:17.0%。tax rebate rate:9.0%。MFN tariff:5.5%。general tariff:30.0% |
|
A new hydroxychavicol dimer from the roots of Piper betle.
Molecules 18(3) , 2563-70, (2013) A new hydroxychavicol dimer, 2-(g'-hydroxychavicol)-hydroxychavicol (1), was isolated from the roots of Piper betle Linn. along with five known compounds, hydroxychavicol (2), aristololactam A II (3),... |
|
|
Piper betel leaf extract: anticancer benefits and bio-guided fractionation to identify active principles for prostate cancer management.
Carcinogenesis 34(7) , 1558-66, (2013) Plant extracts, a concoction of bioactive non-nutrient phytochemicals, have long served as the most significant source of new leads for anticancer drug development. Explored for their unique medicinal... |
|
|
Evaluation of the antimicrobial, antioxidant, and anti-inflammatory activities of hydroxychavicol for its potential use as an oral care agent.
Antimicrob. Agents Chemother. 53 , 216-22, (2009) Hydroxychavicol isolated from the chloroform extraction of aqueous extract of Piper betle leaves showed inhibitory activity against oral cavity pathogens. It exhibited an inhibitory effect on all of t... |
| 3,4-dihydroxy-allylbenzene |
| 4-prop-2-enylbenzene-1,2-diol |
| 4-allylcatechol |
| 4-allylbenzene-1,2-diol |
| 4-Allylpyrocatechol |
| 4-Allyl-1,2-benzenediol |
| 1,2-Benzenediol, 4-(2-propen-1-yl)- |
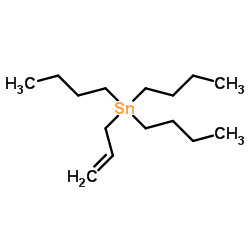
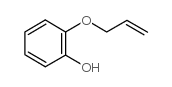
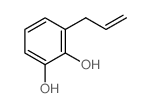
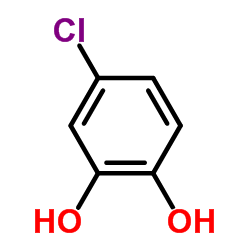
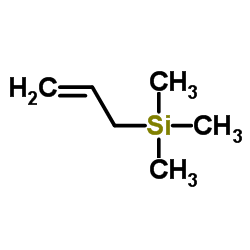

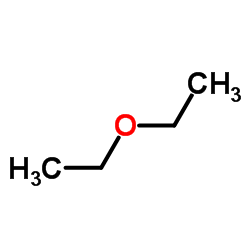
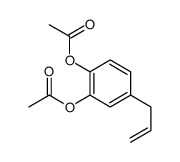 CAS#:13620-82-1
CAS#:13620-82-1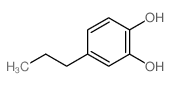 CAS#:2525-02-2
CAS#:2525-02-2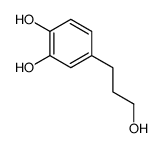 CAS#:46118-02-9
CAS#:46118-02-9 CAS#:5707-55-1
CAS#:5707-55-1 CAS#:62600-15-1
CAS#:62600-15-1