teriflunomide
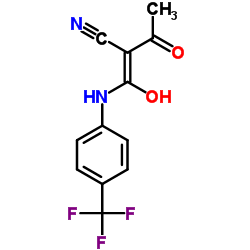
teriflunomide structure
|
Common Name | teriflunomide | ||
|---|---|---|---|---|
| CAS Number | 108605-62-5 | Molecular Weight | 270.207 | |
| Density | 1.4±0.1 g/cm3 | Boiling Point | 363.0±42.0 °C at 760 mmHg | |
| Molecular Formula | C12H9F3N2O2 | Melting Point | N/A | |
| MSDS | USA | Flash Point | 173.3±27.9 °C | |
| Symbol |

GHS07 |
Signal Word | Warning | |
Use of teriflunomide(E/Z)-Teriflunomide ((E/Z)-A77 1726) is the active metabolite of Leflunomide (HY-B0083). Leflunomide is an immunomodulatory agent that may exert effects by inhibiting the mitochondrial enzyme dihydroorotate dehydrogenase (DHODH). Leflunomide can be used for the research of rheumatoid arthritis (RA)[1]. |
| Name | 2-Cyano-3-hydroxy-N-(4-(trifluoromethyl)phenyl)but-2-enamide,teriflunomide |
|---|---|
| Synonym | More Synonyms |
| Description | (E/Z)-Teriflunomide ((E/Z)-A77 1726) is the active metabolite of Leflunomide (HY-B0083). Leflunomide is an immunomodulatory agent that may exert effects by inhibiting the mitochondrial enzyme dihydroorotate dehydrogenase (DHODH). Leflunomide can be used for the research of rheumatoid arthritis (RA)[1]. |
|---|---|
| Related Catalog |
| Density | 1.4±0.1 g/cm3 |
|---|---|
| Boiling Point | 363.0±42.0 °C at 760 mmHg |
| Molecular Formula | C12H9F3N2O2 |
| Molecular Weight | 270.207 |
| Flash Point | 173.3±27.9 °C |
| Exact Mass | 270.061615 |
| PSA | 73.12000 |
| LogP | 0.71 |
| Vapour Pressure | 0.0±0.9 mmHg at 25°C |
| Index of Refraction | 1.552 |
| Storage condition | +2C to +8C |
|
Hepatic cytochrome P450s attenuate the cytotoxicity induced by leflunomide and its active metabolite A77 1726 in primary cultured rat hepatocytes.
Toxicol. Sci. 122(2) , 579-86, (2011) The Black Box Warning section of the U.S. drug label for leflunomide was recently updated to include stronger warnings about potential hepatotoxicity from this novel anti-arthritis drug. Because metab... |
|
|
Respiratory syncytial virus inhibits lung epithelial Na+ channels by up-regulating inducible nitric-oxide synthase.
J. Biol. Chem. 284(11) , 7294-306, (2009) Respiratory syncytial virus (RSV) infection has been shown to reduce Na+-driven alveolar fluid clearance in BALB/c mice in vivo. To investigate the cellular mechanisms by which RSV inhibits amiloride-... |
|
|
Induction of EMT-like phenotypes by an active metabolite of leflunomide and its contribution to pulmonary fibrosis.
Cell Death Differ. 17(12) , 1882-95, (2010) Drug-induced interstitial lung disease (ILD), particularly pulmonary fibrosis, is a serious clinical concern and myofibroblasts have been suggested to have a major role, with it recently being reveale... |
| 2-Butenamide (2-cyano-3-hydroxy-N-[4-(trifluoromethyl)phenyl] |
| Butanenitrile, 2-[hydroxy[[4-(trifluoromethyl)phenyl]amino]methylene]-3-oxo-, (2Z)- |
| (2Z)-2-(Hydroxy{[4-(trifluoromethyl)phenyl]amino}methylene)-3-oxobutanenitrile |
| 2-Cyano-3-hydroxy-N-(4-trifluoromethylphenyl)crotonamide |
| Teriflunomide |
| 2-Cyano-3-hydroxy-N-(4-(trifluoromethyl)phenyl)but-2-enamide |

