Ethyl heptanoate
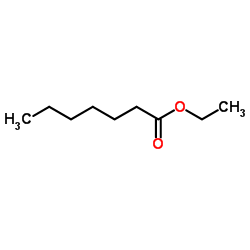
Ethyl heptanoate structure
|
Common Name | Ethyl heptanoate | ||
|---|---|---|---|---|
| CAS Number | 106-30-9 | Molecular Weight | 158.238 | |
| Density | 0.9±0.1 g/cm3 | Boiling Point | 188.3±3.0 °C at 760 mmHg | |
| Molecular Formula | C9H18O2 | Melting Point | −66 °C(lit.) | |
| MSDS | Chinese USA | Flash Point | 66.1±0.0 °C | |
Use of Ethyl heptanoateEthyl heptanoate is a biochemical reagent that can be used as a biological material or organic compound for life science related research. |
| Name | ethyl heptanoate |
|---|---|
| Synonym | More Synonyms |
| Description | Ethyl heptanoate is a biochemical reagent that can be used as a biological material or organic compound for life science related research. |
|---|---|
| Related Catalog |
| Density | 0.9±0.1 g/cm3 |
|---|---|
| Boiling Point | 188.3±3.0 °C at 760 mmHg |
| Melting Point | −66 °C(lit.) |
| Molecular Formula | C9H18O2 |
| Molecular Weight | 158.238 |
| Flash Point | 66.1±0.0 °C |
| Exact Mass | 158.130676 |
| PSA | 26.30000 |
| LogP | 3.37 |
| Vapour Pressure | 0.6±0.4 mmHg at 25°C |
| Index of Refraction | 1.418 |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Personal Protective Equipment | Eyeshields;Gloves;half-mask respirator (US);multi-purpose combination respirator cartridge (US) |
|---|---|
| Hazard Codes | Xi:Irritant; |
| Risk Phrases | R36/37/38 |
| Safety Phrases | S37/39-S26 |
| RIDADR | UN 1993 / PGIII |
| WGK Germany | 1 |
| RTECS | MJ2087000 |
| HS Code | 29159080 |
| Precursor 10 | |
|---|---|
| DownStream 10 | |
| HS Code | 2915900090 |
|---|---|
| Summary | 2915900090 other saturated acyclic monocarboxylic acids and their anhydrides, halides, peroxides and peroxyacids; their halogenated, sulphonated, nitrated or nitrosated derivatives VAT:17.0% Tax rebate rate:9.0% Supervision conditions:AB(certificate of inspection for goods inward,certificate of inspection for goods outward) MFN tariff:5.5% General tariff:30.0% |
|
Effects of droplet crystallization and melting on the aroma release properties of a model oil-in-water emulsion.
J. Agric. Food Chem. 54(5) , 1829-37, (2006) Aroma compounds partition between the dispersed and the continuous phases in emulsions, and phase transitions in the lipid droplets profoundly affect the position of the equilibrium. In the present st... |
|
|
Influence of poly (lactide-co-glycolide) type and gamma irradiation on the betamethasone acetate release from the in situ forming systems.
Curr. Drug Deliv. 6(2) , 184-91, (2009) In situ forming biodegradable polymeric systems were prepared from Poly (DL-lactide-co-glycolide), RG504H (50:50, lactide:glycolide), RG756 (75:25) and mixture of them. They were dissolved in N-methyl... |
|
|
Identification of electrophysiologically-active compounds for the malaria mosquito, Anopheles gambiae, in human sweat extracts.
Med. Vet. Entomol. 10(3) , 269-76, (1996) Human sweat samples were chemically fractionated into acid and non-acid components. The most abundant volatile compounds present in the fractions were identified by linked gas chromatography mass spec... |
| Ethyl heptoate |
| Ethyl heptanoate |
| Grape oil |
| Ethylheptanoat |
| ethyl enanthoate |
| ethyl n-heptanoate |
| Oenanthic ether |
| ethyl oenanthate |
| Enanthylic ether |
| Ethyl enanthate |
| Aether Oenanthieus |
| Cognac Oil Synthetic |
| heptanoic acid ethyl ester |
| Cognac oil |
| Enanthic Acid Ethyl Ester |
| EINECS 203-382-9 |
| MFCD00009538 |
| Ethyl n-Heptoate |
| Aether oenanthicus |
| Heptanoic acid, ethyl ester |
| ethyl heptylate |
| Oil of Grapes |
 CAS#:64-17-5
CAS#:64-17-5 CAS#:111-26-2
CAS#:111-26-2 CAS#:201230-82-2
CAS#:201230-82-2 CAS#:25118-23-4
CAS#:25118-23-4 CAS#:2351-88-4
CAS#:2351-88-4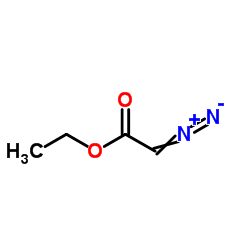 CAS#:623-73-4
CAS#:623-73-4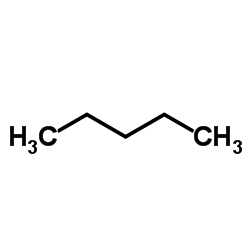 CAS#:109-66-0
CAS#:109-66-0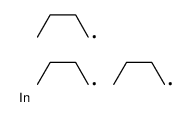 CAS#:15676-66-1
CAS#:15676-66-1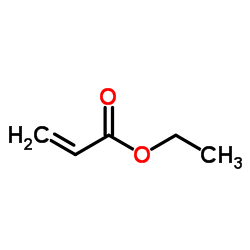 CAS#:140-88-5
CAS#:140-88-5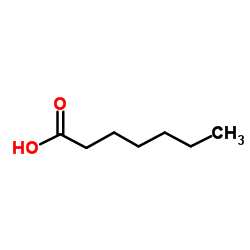 CAS#:111-14-8
CAS#:111-14-8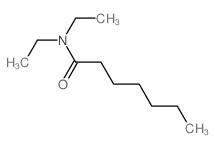 CAS#:10385-08-7
CAS#:10385-08-7 CAS#:373-49-9
CAS#:373-49-9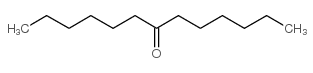 CAS#:462-18-0
CAS#:462-18-0 CAS#:18229-17-9
CAS#:18229-17-9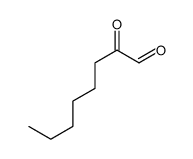 CAS#:2363-86-2
CAS#:2363-86-2 CAS#:111-71-7
CAS#:111-71-7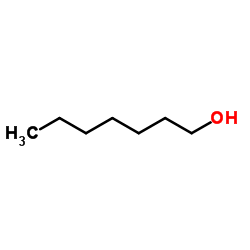 CAS#:111-70-6
CAS#:111-70-6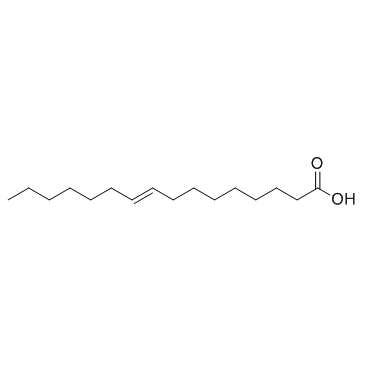 CAS#:10030-73-6
CAS#:10030-73-6
