Phenylthiocarbamide
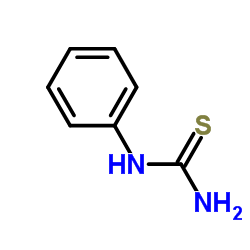
Phenylthiocarbamide structure
|
Common Name | Phenylthiocarbamide | ||
|---|---|---|---|---|
| CAS Number | 103-85-5 | Molecular Weight | 152.217 | |
| Density | 1.3±0.1 g/cm3 | Boiling Point | 266.7±23.0 °C at 760 mmHg | |
| Molecular Formula | C7H8N2S | Melting Point | 145-150 °C(lit.) | |
| MSDS | Chinese USA | Flash Point | 115.1±22.6 °C | |
| Symbol |

GHS06 |
Signal Word | Danger | |
Use of PhenylthiocarbamidePhenylthiourea (Phenylthiocarbamide) is an inhibitor of phenoloxidase. Phenylthiourea inhibits enzymatic oxidation of DOPA by phenoloxidase (Ki: 0.21?μM)[1]. |
| Name | N-phenylthiourea |
|---|---|
| Synonym | More Synonyms |
| Description | Phenylthiourea (Phenylthiocarbamide) is an inhibitor of phenoloxidase. Phenylthiourea inhibits enzymatic oxidation of DOPA by phenoloxidase (Ki: 0.21?μM)[1]. |
|---|---|
| Related Catalog | |
| Target |
phenoloxidase[1] |
| References |
| Density | 1.3±0.1 g/cm3 |
|---|---|
| Boiling Point | 266.7±23.0 °C at 760 mmHg |
| Melting Point | 145-150 °C(lit.) |
| Molecular Formula | C7H8N2S |
| Molecular Weight | 152.217 |
| Flash Point | 115.1±22.6 °C |
| Exact Mass | 152.040817 |
| PSA | 70.14000 |
| LogP | 0.73 |
| Vapour Pressure | 0.0±0.5 mmHg at 25°C |
| Index of Refraction | 1.725 |
| Storage condition | Poison room |
| Stability | Stable. Incompatible with strong acids, strong oxidizing agents, strong bases. |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
MUTATION DATA
|
| Symbol |

GHS06 |
|---|---|
| Signal Word | Danger |
| Hazard Statements | H300-H317 |
| Precautionary Statements | P264-P280-P301 + P310 |
| Personal Protective Equipment | Eyeshields;Faceshields;full-face particle respirator type N100 (US);Gloves;respirator cartridge type N100 (US);type P1 (EN143) respirator filter;type P3 (EN 143) respirator cartridges |
| Hazard Codes | T+ |
| Risk Phrases | R28 |
| Safety Phrases | S28-S36/37-S45 |
| RIDADR | UN 2811 6.1/PG 1 |
| WGK Germany | 3 |
| RTECS | YU1400000 |
| Packaging Group | I |
| Hazard Class | 6.1 |
| HS Code | 2930909090 |
| Precursor 7 | |
|---|---|
| DownStream 8 | |
| HS Code | 2930909090 |
|---|---|
| Summary | 2930909090. other organo-sulphur compounds. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:30.0% |
|
Tweedle cuticular protein BmCPT1 is involved in innate immunity by participating in recognition of Escherichia coli.
Insect Biochem. Mol. Biol. 58 , 76-88, (2015) Bombyx mori, a lepidopteran insect, is one of the earliest models for pattern recognition of Gram-negative bacteria, which may induce the IMD pathway for production of antibacterial peptides. So far, ... |
|
|
Cellular Anti-Melanogenic Effects of a Euryale ferox Seed Extract Ethyl Acetate Fraction via the Lysosomal Degradation Machinery.
Int. J. Mol. Sci. 16 , 9217-35, (2015) The aim of this study was to investigate the effect of ethyl acetate fraction of Euryale ferox seed extracts (Efse-EA) on melanogenesis in immortalized mouse melanocyte cell line, melan-a. Efse-EA sho... |
|
|
Dietary cholesterol directly induces acute inflammasome-dependent intestinal inflammation.
Nat. Commun. 5 , 5864, (2014) Prolonged ingestion of a cholesterol- or saturated fatty acid-enriched diet induces chronic, often systemic, auto-inflammatory responses resulting in significant health problems worldwide. In vivo inf... |
| N′-phenylthiourea |
| N-phenylthiourea |
| PTU |
| 1-Phenylthiourea |
| EINECS 203-151-2 |
| Thiourea, N-phenyl- |
| MFCD00004933 |
| Phenylthiocarbamide |
| 1-PHENYL-2-THIOUREA |
| Phenylthiourea |
| PTC |
 CAS#:103-72-0
CAS#:103-72-0 CAS#:591-50-4
CAS#:591-50-4![Benzamide, N-[(phenylamino)thioxomethyl]- Structure](https://image.chemsrc.com/caspic/158/4921-82-8.png) CAS#:4921-82-8
CAS#:4921-82-8 CAS#:102-08-9
CAS#:102-08-9 CAS#:142-04-1
CAS#:142-04-1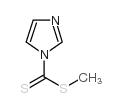 CAS#:74734-11-5
CAS#:74734-11-5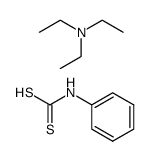 CAS#:43009-16-1
CAS#:43009-16-1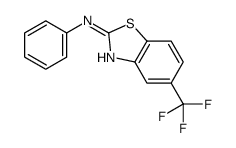 CAS#:105202-01-5
CAS#:105202-01-5 CAS#:615-21-4
CAS#:615-21-4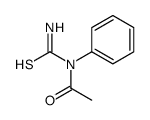 CAS#:22713-55-9
CAS#:22713-55-9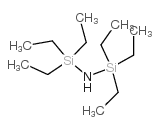 CAS#:2117-18-2
CAS#:2117-18-2 CAS#:994-49-0
CAS#:994-49-0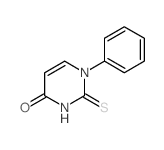 CAS#:21320-99-0
CAS#:21320-99-0
