Levetiracetam

Levetiracetam structure
|
Common Name | Levetiracetam | ||
|---|---|---|---|---|
| CAS Number | 102767-28-2 | Molecular Weight | 170.209 | |
| Density | 1.2±0.1 g/cm3 | Boiling Point | 395.9±25.0 °C at 760 mmHg | |
| Molecular Formula | C8H14N2O2 | Melting Point | 118-119°C | |
| MSDS | Chinese USA | Flash Point | 193.2±23.2 °C | |
| Symbol |

GHS07 |
Signal Word | Warning | |
Use of LevetiracetamLevetiracetam(UCB L059) is a novel anticonvulsant with antihyperalgesic efficacy in inflammatory pain.Target: Calcium ChannelLevetiracetam is used to control some types of seizures in patients with epilepsy. This medicine cannot cure epilepsy and will only work to control seizures for as long as you continue to use it. The exact mechanism for levetiracetam is unknown. However, the drug binds to a synaptic vesicle protein, SV2A, which is believed to impede nerve conduction across synapses [1].Levetiracetam (10-200 mg/kg), ibuprofen (12.5-100 mg/kg), celecoxib (3.75-30 mg/kg), paracetamol (50-200 mg/kg), caffeine (15-100 mg/kg), and 2-drug combinations of levetiracetam with analgesics/caffeine produced a significant, dose-dependent reduction of inflammatory hyperalgesia. Isobolographic analysis revealed that levetiracetam exerts a synergistic interaction with analgesics, with approximately 7-, 9-, and 11-fold reduction of doses of both drugs in combination of levetiracetam with paracetamol, celecoxib, and ibuprofen, respectively. Analysis of the log dose-response curves for levetiracetam (1-50 mg/kg) in the presence of caffeine (10 mg/kg) and levetiracetam applied alone also revealed a synergistic interaction. Levetiracetam's ED50 in the presence of caffeine was reduced approximately 11-fold [2].Clinical indications: Epilepsy; Social phobiaFDA Approved Date: November 2008Toxicity: depression; hallucinations; suicidal thoughts |
| Name | levetiracetam |
|---|---|
| Synonym | More Synonyms |
| Description | Levetiracetam(UCB L059) is a novel anticonvulsant with antihyperalgesic efficacy in inflammatory pain.Target: Calcium ChannelLevetiracetam is used to control some types of seizures in patients with epilepsy. This medicine cannot cure epilepsy and will only work to control seizures for as long as you continue to use it. The exact mechanism for levetiracetam is unknown. However, the drug binds to a synaptic vesicle protein, SV2A, which is believed to impede nerve conduction across synapses [1].Levetiracetam (10-200 mg/kg), ibuprofen (12.5-100 mg/kg), celecoxib (3.75-30 mg/kg), paracetamol (50-200 mg/kg), caffeine (15-100 mg/kg), and 2-drug combinations of levetiracetam with analgesics/caffeine produced a significant, dose-dependent reduction of inflammatory hyperalgesia. Isobolographic analysis revealed that levetiracetam exerts a synergistic interaction with analgesics, with approximately 7-, 9-, and 11-fold reduction of doses of both drugs in combination of levetiracetam with paracetamol, celecoxib, and ibuprofen, respectively. Analysis of the log dose-response curves for levetiracetam (1-50 mg/kg) in the presence of caffeine (10 mg/kg) and levetiracetam applied alone also revealed a synergistic interaction. Levetiracetam's ED50 in the presence of caffeine was reduced approximately 11-fold [2].Clinical indications: Epilepsy; Social phobiaFDA Approved Date: November 2008Toxicity: depression; hallucinations; suicidal thoughts |
|---|---|
| Related Catalog | |
| References |
| Density | 1.2±0.1 g/cm3 |
|---|---|
| Boiling Point | 395.9±25.0 °C at 760 mmHg |
| Melting Point | 118-119°C |
| Molecular Formula | C8H14N2O2 |
| Molecular Weight | 170.209 |
| Flash Point | 193.2±23.2 °C |
| Exact Mass | 170.105530 |
| PSA | 63.40000 |
| LogP | -0.67 |
| Vapour Pressure | 0.0±0.9 mmHg at 25°C |
| Index of Refraction | 1.519 |
| Symbol |

GHS07 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H302-H319 |
| Precautionary Statements | P305 + P351 + P338 |
| Personal Protective Equipment | dust mask type N95 (US);Eyeshields;Gloves |
| Hazard Codes | Xn: Harmful; |
| Risk Phrases | R22 |
| Safety Phrases | S26 |
| RIDADR | NONH for all modes of transport |
| RTECS | UX9656166 |
| HS Code | 2933990090 |
| Precursor 8 | |
|---|---|
| DownStream 1 | |
| HS Code | 2933790090 |
|---|---|
| Summary | 2933790090. other lactams. VAT:17.0%. Tax rebate rate:9.0%. . MFN tariff:9.0%. General tariff:20.0% |
|
Development of a SPE-HPLC-MS/MS method for the determination of most prescribed pharmaceuticals and related metabolites in urban sewage samples.
J. Chromatogr. B. Analyt. Technol. Biomed. Life Sci. 990 , 23-30, (2015) Based on regional prescription data several pharmaceuticals with variable amounts of prescription and corresponding metabolites were selected and analyzed in influent and effluent samples of the sewag... |
|
|
Determining the polymer threshold amount for achieving robust drug release from HPMC and HPC matrix tablets containing a high-dose BCS class I model drug: in vitro and in vivo studies.
AAPS PharmSciTech 16(2) , 398-406, (2015) It is challenging to achieve mechanically robust drug-release profiles from hydrophilic matrices containing a high dose of a drug with good solubility. However, a mechanically robust drug release over... |
|
|
Determination of a selection of anti-epileptic drugs and two active metabolites in whole blood by reversed phase UPLC-MS/MS and some examples of application of the method in forensic toxicology cases.
Drug Test. Anal. 7 , 634-44, (2015) Quantitative determination of anti-epileptic drug concentrations is of great importance in forensic toxicology cases. Although the drugs are not usually abused, they are important post-mortem cases wh... |
| (2S)-2-(2-Oxopyrrolidin-1-yl)butanamide |
| levetiracetamum |
| 2(S)-(2-oxopyrrolidin-1-yl)butyramide |
| KEPPRA |
| (2S)-2-(2-Oxo-1-pyrrolidinyl)butanamide |
| (S)-Etiracetam |
| Etiracetam levo-isomer |
| EINECS 200-659-6 |
| ucb L059 |
| (aS)-a-Ethyl-2-oxo-1-pyrrolidineacetamide |
| 1-pyrrolidineacetamide, a-ethyl-2-oxo-, (aS)- |
| 1-Pyrrolidineacetamide, α-ethyl-2-oxo-, (αS)- |
| (S)-2-(2-Oxopyrrolidin-1-yl)Butanamide |
| SIB S1 |
| (S)-2-(2-Oxo-1-pyrrolidinyl)butyramide |
| MFCD03265610 |
| SIB-S1 |
| (-)-(S)-α-Ethyl-2-oxo-1-pyrrolidineacetamide |
| Keppra XR |
| Levetiracetam |
 CAS#:102849-49-0
CAS#:102849-49-0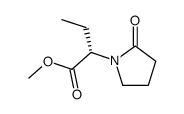 CAS#:358629-51-3
CAS#:358629-51-3 CAS#:332-77-4
CAS#:332-77-4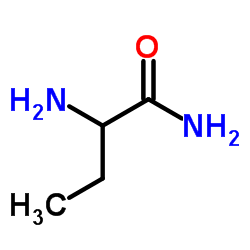 CAS#:7324-11-0
CAS#:7324-11-0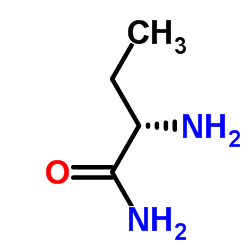 CAS#:53726-14-0
CAS#:53726-14-0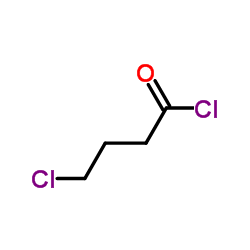 CAS#:4635-59-0
CAS#:4635-59-0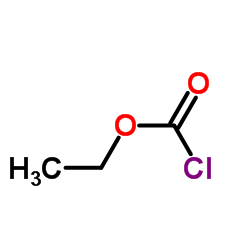 CAS#:541-41-3
CAS#:541-41-3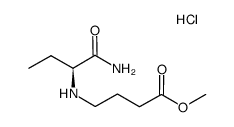 CAS#:429673-91-6
CAS#:429673-91-6 CAS#:664304-29-4
CAS#:664304-29-4