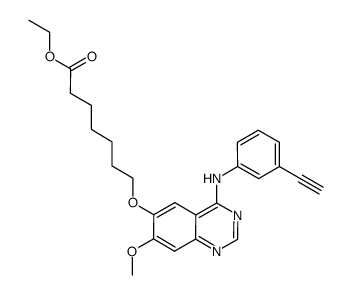
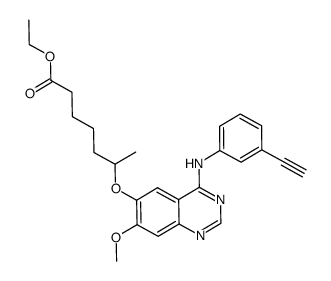
CUDC-101
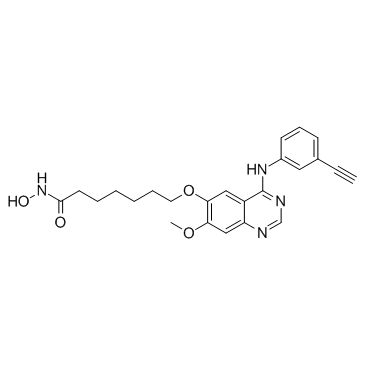
CUDC-101 structure
|
Common Name | CUDC-101 | ||
|---|---|---|---|---|
| CAS Number | 1012054-59-9 | Molecular Weight | 434.488 | |
| Density | 1.3±0.1 g/cm3 | Boiling Point | N/A | |
| Molecular Formula | C24H26N4O4 | Melting Point | 174-177ºC | |
| MSDS | Chinese USA | Flash Point | N/A | |
Use of CUDC-101CUDC-101 is a potent inhibitor of HDAC, EGFR, and HER2 with IC50s of 4.4, 2.4, and 15.7 nM, respectively. |
| Name | 7-[4-(3-ethynylanilino)-7-methoxyquinazolin-6-yl]oxy-N-hydroxyheptanamide |
|---|---|
| Synonym | More Synonyms |
| Description | CUDC-101 is a potent inhibitor of HDAC, EGFR, and HER2 with IC50s of 4.4, 2.4, and 15.7 nM, respectively. |
|---|---|
| Related Catalog | |
| Target |
EGFR:2.4 nM (IC50) HER2:15.7 nM (IC50) HDAC:4.4 nM (IC50) HDAC1:4.5 nM (IC50) HDAC2:12.6 nM (IC50) HDAC3:9.1 nM (IC50) HDAC4:13.2 nM (IC50) HDAC6:5.1 nM (IC50) HDAC5:11.4 nM (IC50) HDAC9:67.2 nM (IC50) HDAC10:26.1 nM (IC50) HDAC8:79.8 nM (IC50) HDAC7:373 nM (IC50) |
| In Vitro | CUDC-101 inhibits both class I and class II HDACs, but not class III, Sir-type HDACs. CUDC-101 displays broad antiproliferative activity in many human cancer cell types. CUDC-101 is a potent and selective HDAC, EGFR, and HER2 inhibitor with only weak inhibition of the following protein kinases (IC50): KDR (VEGFR2) (849 nM), Src (11000 nM), Lyn (840 nM), Lck (5910 nM), Abl-1 (2890 nM), FGFR-2 (3430 nM), Flt-3 (1500 nM), and Ret (3200 nM)[1]. CUDC-101 (300 nM) inhibits both the full length AR (flAR) and the AR variant AR-V7[2]. CUDC-101 is the most active agent in all three ATC cell lines screened for inhibitors of EGFR and HDACs, with half-maximal inhibitory concentration (IC50) at 0.15 μM for 8505c, and 1.66 μM for both C-643 and SW-1736 cells. CUDC-101 inhibits cancer cell migration and modulates epithelial-mesenchymal transition marker expression in ATC cells. CUDC-101 also inhibits HDAC and MAPK pathway, induces p21, and decreases survivin and XIAP expression in ATC cells[3]. CUDC-101 (1 μM) increases the acetylation of p53 and α-tubulin, nonhistone substrates of HDAC, in treated cancer cells. CUDC-101 modulates RTK activity and expression and exhibits immediate and stable inhibition of RTK and downstream Akt signaling[4]. |
| In Vivo | CUDC-101 (120 mg/kg, iv, daily) induces tumor regression in the Hep-G2 liver cancer model and is more efficacious than erlotinib at its maximum tolerated dose (MTD). In the erlotinib-resistant A549 NSCLC xenograft model, CUDC-101 (120 mg/kg) shows potent inhibition of tumor growth. In the erlotinib-sensitive H358 NSCLC models, CUDC-101 (15, 30, 60 mg/kg, i.v.) inhibits tumor growth in a dose-dependent manner. CUDC-101 (120 mg/kg) causes significant tumor regression in the lapatinib-resistant, HER2-negative, EGFR-overexpressing MDA-MB-468 breast cancer model and the EGFR-overexpressing CAL-27 head and neck squamous cell carcinoma (HNSCC) model. CUDC-101 (120 mg/kg) also inhibits tumor growth in the K-ras mutant HCT116 colorectal and EGFR/HER2 (neu)-expressing HPAC pancreatic cancer models[1]. In an in vivo mouse model of metastatic ATC, CUDC-101 inhibits tumor growth and metastases, and significantly prolongs survival[3]. CUDC-101 (120 mg/kg) is effective against a broad range of tumor types in xenograft models[4]. |
| Kinase Assay | The activities of Class I and II HDACs are assessed using the Biomol Color de Lys system. Briefly, HeLa cell nuclear extracts are used as a source of HDACs. Different concentrations of drugs are added to HeLa cell nuclear extracts in the presence of a colorimetric artificial substrate. Developer is added at the end of the assay and enzyme activity is measured in the Wallac Victor II 1420 microplate reader at 405 nM. |
| Cell Assay | Cancer cell lines are plated at 5000 to 10 000 cells per well in 96-well flat-bottomed plates with varying concentrations of compounds. The cells are incubated with compounds for 72 h in the presence of 0.5% of fetal bovine serum. Growth inhibition is assessed by an adenosine triphosphate (ATP) content assay using the Perkin-Elmer ATPlite kit. |
| Animal Admin | Four- to six-week-old female athymic mice (nude nu/nu CD-1) are inoculated subcutaneously into the right hind flank region with 1 to 5×106 cells in a medium suspension of 100−200 μL. For orthotopic implantation of breast cancer cells, a cell suspension in 100 μL of medium is injected directly into the mammary fat pads through a 27G needle. Different doses of CUDC-101, standard anticancer agents and vehicle are administered orally, intraperitoneally, or via tail vein injection as indicated. |
| References |
| Density | 1.3±0.1 g/cm3 |
|---|---|
| Melting Point | 174-177ºC |
| Molecular Formula | C24H26N4O4 |
| Molecular Weight | 434.488 |
| Exact Mass | 434.195404 |
| PSA | 109.09000 |
| LogP | 2.84 |
| Appearance of Characters | white |
| Index of Refraction | 1.638 |
| Storage condition | ?20°C |
| Water Solubility | DMSO: soluble |
| RIDADR | NONH for all modes of transport |
|---|
|
~83% 
CUDC-101 CAS#:1012054-59-9 |
| Literature: Cai, Xiong; Zhai, Hai-Xiao; Wang, Jing; Forrester, Jeffrey; Qu, Hui; Yin, Ling; Lai, Cheng-Jung; Bao, Rudi; Qian, Changgeng Journal of Medicinal Chemistry, 2010 , vol. 53, # 5 p. 2000 - 2009 |
|
~41% 
CUDC-101 CAS#:1012054-59-9 |
| Literature: Cai, Xiong; Qian, Changgeng; Zhai, Haixiao Patent: US2009/76022 A1, 2009 ; Location in patent: Page/Page column 68 ; US 20090076022 A1 |
|
Discovery of 7-(4-(3-ethynylphenylamino)-7-methoxyquinazolin-6-yloxy)-N-hydroxyheptanamide (CUDc-101) as a potent multi-acting HDAC, EGFR, and HER2 inhibitor for the treatment of cancer.
J. Med. Chem. 53 , 2000-2009, (2010) By incorporating histone deacetylase (HDAC) inhibitory functionality into the pharmacophore of the epidermal growth factor receptor (EGFR) and human epidermal growth factor receptor 2 (HER2) inhibitor... |
|
|
CUDC-101, a multitargeted inhibitor of histone deacetylase, epidermal growth factor receptor, and human epidermal growth factor receptor 2, exerts potent anticancer activity.
Cancer Res. 70 , 3647-3656, (2010) Receptor tyrosine kinase inhibitors have recently become important therapeutics for a variety of cancers. However, due to the heterogeneous and dynamic nature of tumors, the effectiveness of these age... |
| 7-({4-[(3-Ethynylphenyl)amino]-7-methoxy-6-quinazolinyl}oxy)-N-hydroxyheptanamide |
| Heptanamide, 7-[[4-[(3-ethynylphenyl)amino]-7-methoxy-6-quinazolinyl]oxy]-N-hydroxy- |
| CUDC 101 |
| UNII-1A7Y9MP123 |
| 7-(4-(3-Ethynylphenylamino)-7-methoxyquinazolin-6-yloxy)-N-hydroxyheptanamide |
| CUDC101 |
| CUDC-101 |
| S1194_Selleck |