56795-51-8
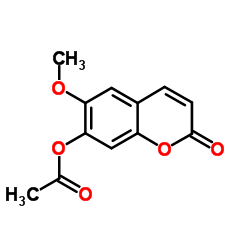
| Name | 6-Methoxy-2-oxo-2H-chromen-7-yl acetate |
|---|---|
| Synonyms |
O-Acetyl-afromosin
Afrormosin-monoacetat 7-acetoxy-6-methoxycoumarin O-Acetyl-afrormosin 7-O-Acetyl-scopoletin Afrormosinacetat 7-O-acetylafrormosin 6-Methoxy-2-oxo-2H-chromen-7-yl acetate Scopoletin-acetat Isoflavone,7-hydroxy-4',6-dimethoxy-,acetate 7-acetoxy-6-methoxy-chromen-2-one 7-Hydroxy-4',6-dimethoxyisoflavone acetate Afromosin-acetat 7-O-acetylafromosin 7-O-Ac-afromosin 7-Acetoxy-6,4'-dimethoxy-isoflavon 7-Acetoxy-6-methoxy-cumarin |
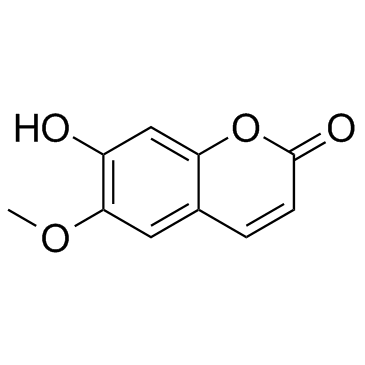
| Description | Scopoletin acetate is a coumarin isolated from Artemisia granatensis[1]. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.3±0.1 g/cm3 |
|---|---|
| Boiling Point | 379.4±42.0 °C at 760 mmHg |
| Molecular Formula | C12H10O5 |
| Molecular Weight | 234.205 |
| Flash Point | 170.5±27.9 °C |
| Exact Mass | 234.052826 |
| PSA | 65.74000 |
| LogP | 0.55 |
| Vapour Pressure | 0.0±0.9 mmHg at 25°C |
| Index of Refraction | 1.560 |
| Hazard Codes | Xi |
|---|---|
| HS Code | 2932209090 |
|
~% 
56795-51-8 |
| Literature: Seka; Kallir Chemische Berichte, 1931 , vol. 64, p. 909,915 Full Text Show Details Head; Alexander Journal of the Chemical Society, 1931 , p. 1241,1244 |
|
~% 
56795-51-8 |
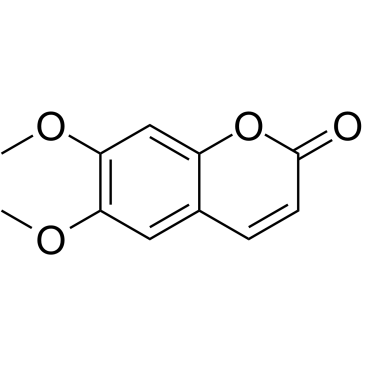
| Literature: Power; Moore Journal of the Chemical Society, 1909 , vol. 95, p. 243 Journal of the Chemical Society, vol. 250, p. 256 |
| Precursor 2 | |
|---|---|
| DownStream 2 | |
| HS Code | 2932209090 |
|---|---|
| Summary | 2932209090. other lactones. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |