18642-44-9
| Name | Actein |
|---|---|
| Synonyms |
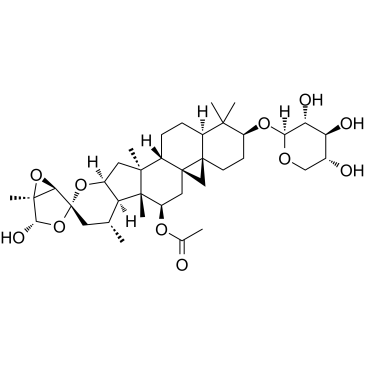
β-D-Xylopyranoside, (1'R,2S,4'S,4aR,5'S,5aR,7R,7aR,7bR,8R,10R,11aS,12aS,12bS,14aR)-7-(acetyloxy)hexadecahydro-4'-hydroxy-1,1,5',7a,8,12a-hexamethylspiro[5H-cyclopropa[1',8'a]naphth[2',1':4,5]indeno [2,1-b]pyran-10(2H),2'-[3,6]dioxabicyclo[3.1.0]hexan]-2-yl
(1'R,2S,4'S,4aR,5'S,5aR,7R,7aR,7bR,8R,10R,11aS,12aS,12bS,14aR)-4'-Hydroxy-1,1,5',7a,8,12a-hexamethyl-2-(β-D-xylopyranosyloxy)hexadecahydro-2H-spiro[cyclopropa[1',8a']naphtho[2',1':4,5]indeno[2,1-b] pyran-10,2'-[3,6]dioxabicyclo[3.1.0]hexan]-7-yl acetate |
| Description | Actein is a triterpene glycoside isolated from the rhizomes of Cimicifuga foetida. Actein suppresses cell proliferation, induces autophagy and apoptosis through promoting ROS/JNK activation, and blunting AKT pathway in human bladder cancer. Actein has little toxicity in vivo[1][2]. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.4±0.1 g/cm3 |
|---|---|
| Melting Point | 246-250ºC |
| Molecular Formula | C37H56O11 |
| Molecular Weight | 676.834 |
| Exact Mass | 676.382263 |
| PSA | 156.67000 |
| LogP | 5.67 |
| Index of Refraction | 1.610 |
| Hazard Codes | Xi |
|---|---|
| RIDADR | NONH for all modes of transport |