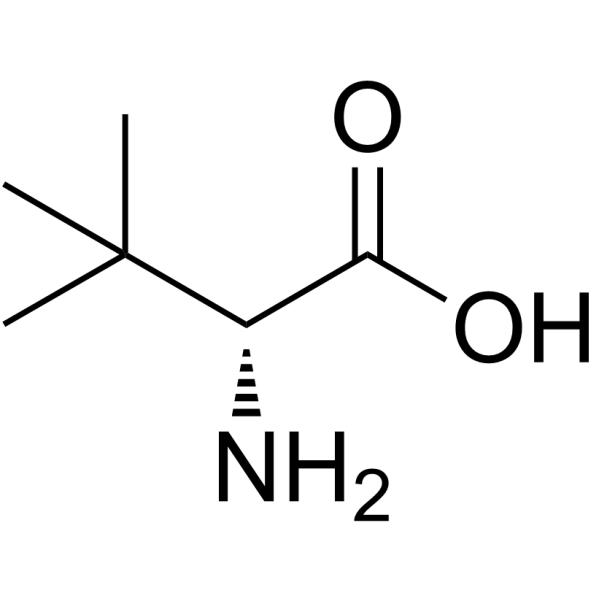
124655-17-0
| Name | (2R)-3,3-dimethyl-2-[(2-methylpropan-2-yl)oxycarbonylamino]butanoic acid |
|---|---|
| Synonyms |
boc-d-tle-oh
(R)-Boc-tert-leucine (R)-2-((tert-Butoxycarbonyl)amino)-3,3-dimethylbutanoic acid MFCD00065575 BOC-D-T-LEU (R)-N-<(1,1-dimethylethoxy)carbonyl>-tert-leucine (R)-2-(N-t-butoxycarbonylamino)-3,3-dimethylbutanoic acid (R)-2-(Boc-amino)-3,3-dimethylbutyric Acid (r)-n-(tert-butoxycarbonyl)-tert-leucine N-(tert-Butoxycarbonyl)-3-methyl-D-valine N-(tert-Butoxycarbonyl)-3-methyl-L-valine N-Boc-D-tert-leucine (R)-2-(tert-Butoxycarbonylamino)-3,3-dimethylbutyric Acid (R)-2-(tert-butoxycarbonylamino)-3,3-dimethylbutanoic acid N-(tert-Butoxycarbonyl)-D-tert-leucine 3-Methyl-N-{[(2-methyl-2-propanyl)oxy]carbonyl}-D-valine N-{[(1,1-dimethylethyl)oxy]carbonyl}-3-methyl-D-valine 3-Methyl-N-{[(2-methyl-2-propanyl)oxy]carbonyl}-L-valine boc-d-tert-leucine (R)-2-tert-butoxycarbonylamino-3,3-dimethylbutyric acid boc-tbu-d-gly-oh |
| Description | Boc-D-tert-leucine is a leucine derivative[1]. |
|---|---|
| Related Catalog | |
| In Vitro | Amino acids and amino acid derivatives have been commercially used as ergogenic supplements. They influence the secretion of anabolic hormones, supply of fuel during exercise, mental performance during stress related tasks and prevent exercise induced muscle damage. They are recognized to be beneficial as ergogenic dietary substances[1]. |
| References |
| Density | 1.1±0.1 g/cm3 |
|---|---|
| Boiling Point | 350.0±25.0 °C at 760 mmHg |
| Melting Point | 118-121ºC |
| Molecular Formula | C11H21NO4 |
| Molecular Weight | 231.289 |
| Flash Point | 165.5±23.2 °C |
| Exact Mass | 231.147064 |
| PSA | 75.63000 |
| LogP | 2.33 |
| Vapour Pressure | 0.0±1.6 mmHg at 25°C |
| Index of Refraction | 1.462 |
| Storage condition | Store at 0-5°C |
|
~83% 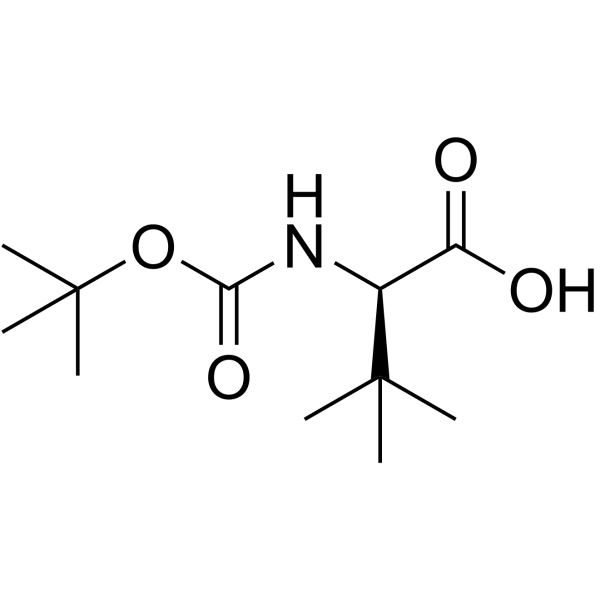
124655-17-0 |
| Literature: GLAXO GROUP LIMITED; ALVARO, Giuseppe; DECOR, Anne; FONTANA, Stefano; HAMPRECHT, Dieter; LARGE, Charles; MARASCO, Agostino Patent: WO2011/69951 A1, 2011 ; Location in patent: Page/Page column 77 ; WO 2011/069951 A1 |
|
~% 
124655-17-0 |
| Literature: Chemical and Pharmaceutical Bulletin, , vol. 40, # 4 p. 1077 - 1079 |
|
~% 
124655-17-0 |
| Literature: Tetrahedron, , vol. 53, # 4 p. 1275 - 1294 |
|
~% 
124655-17-0 |
| Literature: Tetrahedron, , vol. 53, # 4 p. 1275 - 1294 |
|
~% 
124655-17-0 |
| Literature: US2013/66109 A1, ; |
|
~% 
124655-17-0 |
| Literature: Tetrahedron Asymmetry, , vol. 17, # 13 p. 1995 - 1999 |
|
~% 
124655-17-0 |
| Literature: US2013/66109 A1, ; |
| Precursor 4 | |
|---|---|
| DownStream 1 | |