56775-88-3
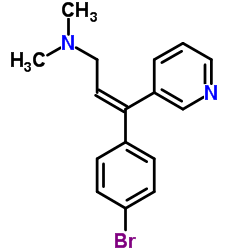
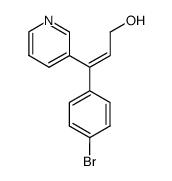
| Name | Zimelidine dihydrochloride,(2Z)-3-(4-Bromophenyl)-N,N-dimethyl-3-(3-pyrindinyl)-2-propen-1-amine |
|---|---|
| Synonyms |
Normud
cis-H-102/09 cis-H 102/09 Hydrochloride,zimeldine (2Z)-3-(4-Bromophenyl)-N,N-dimethyl-3-(pyridin-3-yl)prop-2-en-1-amine H-102/09 Zimeldine Zelmid Zimelidine (Z)-3-(4-Bromophenyl)-N,N-dimethyl-3-(3-pyridinyl)-2-propen-1-amine cis-Zimelidine (2Z)-3-(4-Bromophenyl)-N,N-dimethyl-3-(3-pyridinyl)-2-propen-1-amine |
| Description | Zimelidine is a potent and selective inhibitor of serotonin 5-HT uptake and SERT. Zimelidine is an antidepressant[1][2][3][4]. |
|---|---|
| Related Catalog | |
| In Vivo | Zimelidine (15 mg/kg, IP, once) reduces the development of tolerance to morphine-induced antinociception in rats[3]. Zimelidine (5 mg/kg, IP, daily for 14 days) dose not modify the responsiveness of CA3 hippocampal pyramidal neurons to microiontophoreticahy applied 5-HT[2]. Zimelidine (0.2, 2 and 20 nmol/100 nL) in medial amygdaloid nucleus (MeA) evokes dose dependent hypophagic effects in fasted rats[4]. Animal Model: Male Wistar albino rats (160-180 g, n=72)[3] Dosage: 15 mg/kg Administration: IP, once Result: Significantly attenuated the development and expression of morphine tolerance. The maximal antinociceptive effect of Zimelidine was obtained at the 60 minutes measurements in the zimelidine group and at the 30 minutes measurements in the morphine tolerant group by the tail-flick and hot-plate tests. Administration of zimelidine with morphine showed additive analgesic effect. Animal Model: Sprague-Dawley rats (150 to 250 g, Ten, male)[2] Dosage: 5 mg/kg Administration: IP, daily for 14 days Result: Did not modify the responsiveness of CA3 hippocampal pyramidal neurons to microiontophoreticahy applied 5-HT. |
| References |
| Density | 1.3±0.1 g/cm3 |
|---|---|
| Boiling Point | 412.8±45.0 °C at 760 mmHg |
| Molecular Formula | C16H19BrCl2N2 |
| Molecular Weight | 317.224 |
| Flash Point | 203.4±28.7 °C |
| Exact Mass | 316.057495 |
| PSA | 16.13000 |
| LogP | 4.63 |
| Vapour Pressure | 0.0±1.0 mmHg at 25°C |
| Index of Refraction | 1.597 |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Precursor 10 | |
|---|---|
| DownStream 3 | |