5289-74-7
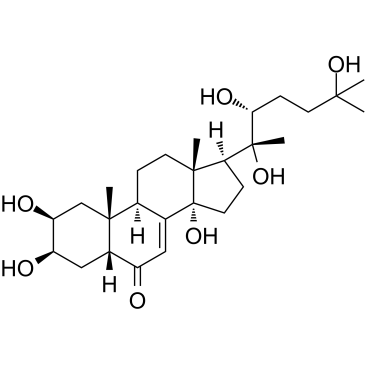
| Name | 20-hydroxyecdysone |
|---|---|
| Synonyms |
CRUSTECDYSONE
20-hydroxyecdysone MFCD00036740 (2S,3R,5R,9R,10R,13R,14S,17S)-2,3,14-Trihydroxy-10,13-dimethyl-17-[(2R,3R)-2,3,6-trihydroxy-6-methyl-2-heptanyl]-1,2,3,4,5,9,10,11,12,13,14,15,16,17-tetradecahydro-6H-cyclopenta[a]phenanthren-6-on β-ecdysone (2β,3β,5β,22R)-2,3,14,20,22,25-Hexahydroxycholest-7-en-6-one Hydroxyecdysone b-Ecdysone Isoinokosterone Commisterone (2b,3b,5b,22R)-2,3,14,20,22,25-Hexahydroxycholest-7-en-6-one Ecdysterone (2S,3R,5R,9R,10R,13R,14S,17S)-2,3,14-Trihydroxy-10,13-dimethyl-17-[(2R,3R)-2,3,6-trihydroxy-6-methyl-2-heptanyl]-1,2,3,4,5,9,10,11,12,13,14,15,16,17-tetradecahydro-6H-cyclopenta[a]phenanthren-6-one |
| Description | Crustecdysone (20-Hydroxyecdysone) is a naturally occurring ecdysteroid hormone isolated from Cyanotis arachnoides C.B.Clarke which controls the ecdysis (moulting) and metamorphosis of arthropods, it inhibits caspase activity and induces autophagy via the 20E nuclear receptor complex, EcR-USP[1].Crustecdysone exhibits regulatory or protective roles in the cardiovascular system[2]. |
|---|---|
| Related Catalog | |
| References |
| Density | 1.3±0.1 g/cm3 |
|---|---|
| Boiling Point | 702.1±60.0 °C at 760 mmHg |
| Melting Point | 242-244 °C |
| Molecular Formula | C27H44O7 |
| Molecular Weight | 480.634 |
| Flash Point | 392.4±29.4 °C |
| Exact Mass | 480.308716 |
| PSA | 138.45000 |
| LogP | -0.53 |
| Vapour Pressure | 0.0±5.0 mmHg at 25°C |
| Index of Refraction | 1.597 |
| Storage condition | 2-8°C |
| Water Solubility | alcohol: soluble |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
MUTATION DATA
|
| Personal Protective Equipment | Eyeshields;Gloves;type N95 (US);type P1 (EN143) respirator filter |
|---|---|
| Hazard Codes | Xi |
| Risk Phrases | R36/37/38 |
| Safety Phrases | S22-S24/25 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 2 |
| RTECS | FZ8060000 |
| Packaging Group | II; III |
| Hazard Class | 4.1 |
| HS Code | 2937290090 |
| HS Code | 2937290090 |
|---|