2203-97-6
| Name | Hydrocortisone 21-hemisuccinate |
|---|---|
| Synonyms |
Hydrocortisone 21-he
MFCD00046256 saxizon cortisol,hydrogensuccinate cortisolsuccinate Butanedioic acid, mono(11,17-dihydroxy-3,20-dioxopregn-4-en-21-yl) ester hydrocortisone hemisuccinate ester 4-{[(11α)-11,17-Dihydroxy-3,20-dioxopregn-4-en-21-yl]oxy}-4-oxobutanoic acid CORTISOL HEMISUCCINATE Hydrocortisone hydrogen succinate 4-[(11,17-Dihydroxy-3,20-dioxopregn-4-en-21-yl)oxy]-4-oxobutanoic acid Hydrocortisone hemisuccinate EINECS 218-612-3 CORTISOL 21-HEMISUCCINATE hydrocortisone-21-O-hemisuccinate Hydrocortizone Succinate Butanedioic acid, mono[(11α)-11,17-dihydroxy-3,20-dioxopregn-4-en-21-yl] ester hydrocortisone21-hemisuccinate*freeacid HYDROCORTISONE SUCCINATE |
| Description | Hydrocortisone hemisuccinate (Hydrocortisone 21-hemisuccinate), a physiological glucocorticoid, and is an orally active steroidal anti-inflammatory drug (SAID). Hydrocortisone hemisuccinate inhibits proinflammatory cytokine activity, with IC50s of 6.7 and 21.4 μM for IL-6 and IL-3, respectively. Hydrocortisone hemisuccinate can be used for the research of ulcerative colitis (UC)[1][2][3]. |
|---|---|
| Related Catalog | |
| Target |
IL-6:6.7 μM (IC50) IL-3:21.4 μM (IC50) |
| In Vitro | Hydrocortisone hemisuccinate inhibits IL-6 and IL-3 bioactivity, with IC50s of 6.7 and 21.4 μM, respectively, and shows no cytotoxic effects on IL-6-independent MH60 cells[3]. Hydrocortisone hemisuccinate (0.12-60 μM; 72 h) inhibits phytohemagglutinin (PHA) response in peripheral lymphocytes (PBL) and T-lymphocytes cultures[3]. |
| In Vivo | Hydrocortisone hemisuccinate (30 mg/kg; p.o. twice daily for 5 d) reduces the weight loss and increases the food intake in mice[2]. Animal Model: Male Sprague-Dawley rats (200-220 g, 10-11 weeks) are induced colitis[2] Dosage: 30 mg/kg Administration: P.o. twice daily for 5 days Result: Significantly decreased the disease activity index (DAI) scores and myeloperoxidase (MPO) activity compared to the 2, 4, 6-trinitrobenzenesulfonic acid (TNBS) group. Increased the body weight. |
| References |
| Density | 1.3±0.1 g/cm3 |
|---|---|
| Boiling Point | 685.5±55.0 °C at 760 mmHg |
| Molecular Formula | C25H34O8 |
| Molecular Weight | 462.533 |
| Flash Point | 231.1±25.0 °C |
| Exact Mass | 462.225372 |
| PSA | 138.20000 |
| LogP | 2.13 |
| Vapour Pressure | 0.0±4.8 mmHg at 25°C |
| Index of Refraction | 1.587 |
| Storage condition | −20°C |
| Water Solubility | Practically insoluble in water, freely soluble in acetone and in anhydrous ethanol. It dissolves in dilute solutions of alkali carbonates and alkali hydroxides. |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Personal Protective Equipment | Eyeshields;Gloves;type N95 (US);type P1 (EN143) respirator filter |
|---|---|
| Hazard Codes | Xi |
| Safety Phrases | S22-S24/25 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| RTECS | TU5010147 |
|
~98% 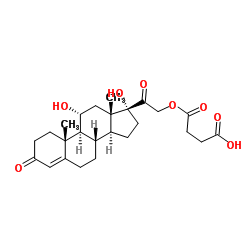
2203-97-6 |
| Literature: Wang, Chao; Zhao, Ming; Qiu, Xuecai; Peng, Shiqi Bioorganic and Medicinal Chemistry, 2004 , vol. 12, # 16 p. 4403 - 4421 |