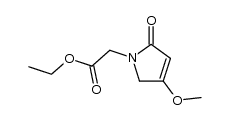
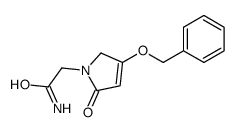
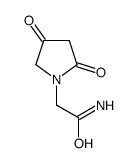
62613-82-5
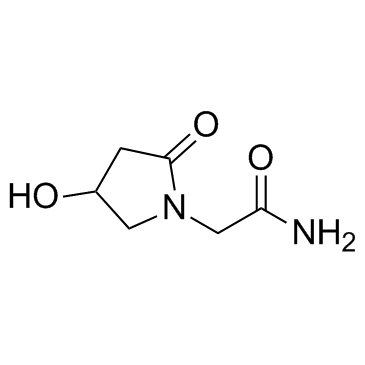
| Name | 4-Hydroxy-2-Oxopyrrolidine-N-Acetamide |
|---|---|
| Synonyms |
4-Hydroxy-2-oxopyrrolidine-N-acetamide
2-(4-Hydroxypyrrolidin-2-on-1-yl)acetamide MFCD00242951 oxiracetam 2-(4-hydroxy-2-oxopyrrolidin-1-yl)acetamide 2-(4-Hydroxy-2-oxo-1-pyrrolidinyl)acetamide UNII:P7U817352G |
| Description | Oxiracetam is a cyclic derivative of γ-aminobutyric acid (GABA) which has been commonly used as nootropic drug to treat cognitive impairments. |
|---|---|
| Related Catalog | |
| Target |
GABA receptor[1] |
| References |
| Density | 1.4±0.1 g/cm3 |
|---|---|
| Boiling Point | 494.6±40.0 °C at 760 mmHg |
| Melting Point | 165-168ºC |
| Molecular Formula | C6H10N2O3 |
| Molecular Weight | 158.155 |
| Flash Point | 252.9±27.3 °C |
| Exact Mass | 158.069138 |
| PSA | 83.63000 |
| LogP | -2.48 |
| Vapour Pressure | 0.0±2.9 mmHg at 25°C |
| Index of Refraction | 1.570 |
| Storage condition | 2-8°C |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Symbol |

GHS07 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H315-H319 |
| Precautionary Statements | P305 + P351 + P338 |
| Personal Protective Equipment | dust mask type N95 (US);Eyeshields;Gloves |
| Hazard Codes | Xi |
| Risk Phrases | R36/38 |
| Safety Phrases | S26-S36 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| RTECS | UX9656638 |
| HS Code | 2933790090 |
| Precursor 8 | |
|---|---|
| DownStream 0 | |
| HS Code | 2933790090 |
|---|---|
| Summary | 2933790090. other lactams. VAT:17.0%. Tax rebate rate:9.0%. . MFN tariff:9.0%. General tariff:20.0% |