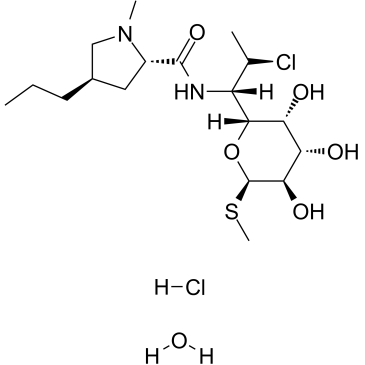
58207-19-5
| Name | Clindamycin Hydrochloride Monohydrate |
|---|---|
| Synonyms |
Methyl (5R)-5-[(1S,2S)-2-chloro-1-{[(4R)-1-methyl-4-propyl-L-prolyl]amino}propyl]-1-thio-β-L-arabinopyranoside hydrochloride hydrate
clindamycin hydrochloride hydrate L-threo-α-D-galacto-Octopyranoside, methyl 7-chloro-6,7,8-trideoxy-6-[[[(2S,4R)-1-methyl-4-propyl-2-pyrrolidinyl]carbonyl]amino]-1-thio-, hydrochloride, hydrate (1:1:1) |
| Description | Clindamycin hydrochloride monohydrate is an oral protein synthesis inhibitory agent that has the ability to suppress the expression of virulence factors in Staphylococcus aureus at sub-inhibitory concentrations (sub-MICs). Clindamycin hydrochloride monohydrate resistance results from enzymatic methylation of the antibiotic binding site in the 50S ribosomal subunit (23S rRNA). Clindamycin hydrochloride monohydrate decreases the production of Panton-Valentine leucocidin (PVL), toxic-shock-staphylococcal toxin (TSST-1) or alpha-haemolysin (Hla)[1]. |
|---|---|
| Related Catalog | |
| References |
| Boiling Point | 647ºC at 760 mmHg |
|---|---|
| Melting Point | 143ºC |
| Molecular Formula | C18H36Cl2N2O6S |
| Molecular Weight | 479.459 |
| Flash Point | 345.1ºC |
| Exact Mass | 478.167114 |
| PSA | 136.79000 |
| LogP | 1.45600 |
| Index of Refraction | 143 ° (C=2, H2O) |
| Water Solubility | Soluble in water |
| Hazard Codes | Xi: Irritant; |
|---|---|
| Risk Phrases | 36/37/38 |
| Safety Phrases | 26-37/39 |
| RIDADR | NONH for all modes of transport |
| RTECS | GF2275000 |
| HS Code | 29419090 |