9041-93-4
| Name | Bleomycin sulfate |
|---|---|
| Synonyms |
Blenmycins
BleoMycin Sufate BLEMOYCIN SULFATE Bleomycin (sulfate) BLENOXANE MFCD00070310 BLEO BLEXANE BLEOMYCIN SULPHATE Blocamicina EINECS 232-925-2 |
| Description | Bleomycin sulfate is a DNA synthesis inhibitor with potent antitumor activity. |
|---|---|
| Related Catalog | |
| Target |
DNA/RNA Synthesis[1] |
| In Vitro | Bleomycin (BLM) is chosen as the best-studied micronucleus (MN) inducers in human lymphocytes with different mechanisms of genotoxicity. The most frequent Bleomycin-induced DNA lesions are single and double strand breaks and single apuinic/apyrimidinic sites. At the same time Bleomycin is true radiomimetic compound, resembling almost completely the genetic effect of ionizing radiation[1]. The IC50 value of Bleomycin sulfate for UT-SCC-19A cell line is 4.0±1.3 nM. UT-SCC-12A and UT-SCC-12B are both more resistant to Bleomycin (BLM); IC50 values are 14.2±2.8 nM and 13.0±1.1 nM, respectively[2]. Bleomycin (BLM) induces a significant increase in the percentage of aberrant cells (i.e., cells showing at least one aberration) and in the frequency of chromosomal aberrations per cell compare with control cultures 18 h after treatment (p<0.05)[3]. |
| In Vivo | A short-range beta-emitting radionuclide combined to Bleomycin (In-111-BLMC) is a tumor-targeting agent in SCCs. Within 35 days the weight of nude mice increases 2.8±0.6g. At 25 and 35 days after tumor inoculations the tumor volumes are 111±51 mm3 and 874±577 mm3, respectively. The calculated doubling time is 3.86±0.76 days. SCC cell lines demonstrate different sensitivity to Bleomycin. Our SCC tumor xenograft model shows a rapid growth proper for radiochemotherapeutic studies using In-111-BLMC. The uptake of In-111-BLMC in vivo has been directly proportional to proliferation activity, and the tumors with high binding capacity could be predicted from animal model dose calculations[2]. At 7 and 14 days after Bleomycin (BLM) treatment, the signal of TGF-β1 is significantly stronger than that of the control group. At 28 days after treatment, the TGF-β1 signal became a little weaker. At 7 and 14 days of Bleomycin plus Dex group, the signal of TGF-β1 is also stronger than that of the control group. However, at 28 days, the TGF-β1 signal become weaker and is a little stronger than the level of control group. All the results are given by comparison of the average IOD value[4]. |
| Cell Assay | ADIPO-P2 cells are grown in D-MEM high glucose medium supplemented with 20% fetal calf serum, penicillin (100 U/mL) and streptomycin (100 μg/mL) at 37°C and 5% CO2 atmosphere. Cells are cultured as monolayer in TC25 Corning flasks containing 1.5×105 cells/mL. For each experiment, two flasks are set up, one for the control and one for the treated culture. During the log phase of growth ADIPO-P2 cells are treated with a 30 min pulse of 2.5 μg/mL of Bleomycin (dissolved in sterile 0.9% NaCl). Control cultures are set up in parallel but not exposed to Bleomycin. At the end of the pulse treatment with Bleomycin, the cells are washed twice with Hank's balanced salt solution and kept in culture with fresh culture medium until harvesting. Cells are continuously maintained in culture during 5 passages or subcultures after treatment. Subcultivation is carried out whenever the cultures became confluent (approximately 4×105 cells/mL of culture medium). To estimate cell growth, at the time of subcultivation cells are collected by trypsinization, an aliquot of about 200 μL stained with 0.4% trypan blue, and the number of viable cells (cells not stained) is determined[3]. |
| Animal Admin | Mice[4] 60 CD-1 mice are randomly divided into the following 3 groups (n=20 each): saline-water; Bleomycin -water; Bleomycin plus Dexamethasone (Dex). Mice in the saline group are injected intratracheally with 2 mL/kg saline; the others are injected intratracheally with Bleomycin (5 mg/kg, 2 mL/kg in saline). Twenty-four hours after Bleomycin treatment, mice are given by gavage 0.45 mg/kg/d DEX. The day of intratracheal injection with Bleomycin or saline is designated day 0. |
| References |
| Melting Point | 197ºC (dec) |
|---|---|
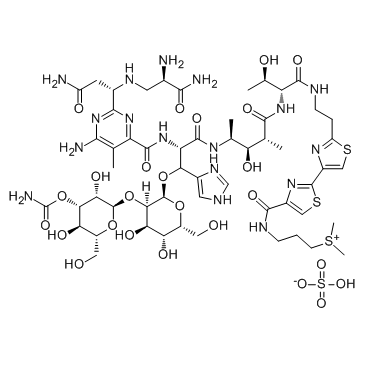
| Molecular Formula | C55H85N17O25S4 |
| Molecular Weight | 1512.62 |
| PSA | 1506.34000 |
| Water Solubility | H2O: 20 mg/mL |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
MUTATION DATA
|
| Symbol |

GHS08 |
|---|---|
| Signal Word | Danger |
| Hazard Statements | H340-H351-H361 |
| Precautionary Statements | P201-P280-P308 + P313 |
| Personal Protective Equipment | Eyeshields;full-face particle respirator type N100 (US);Gloves;respirator cartridge type N100 (US);type P1 (EN143) respirator filter;type P3 (EN 143) respirator cartridges |
| Hazard Codes | T: Toxic;Xi: Irritant; |
| Risk Phrases | R46 |
| Safety Phrases | S53-S36/37-S45 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| RTECS | EC5991990 |