| Structure | Name/CAS No. | Articles |
|---|---|---|
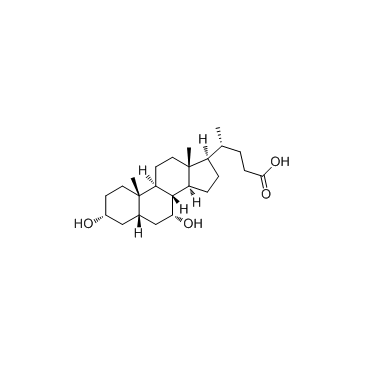 |
Chenodeoxycholic acid
CAS:474-25-9 |
|
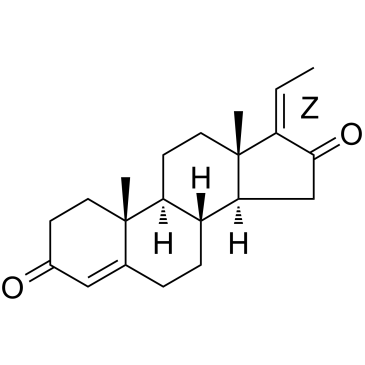 |
Z-Guggulsterone
CAS:39025-23-5 |