| Structure | Name/CAS No. | Articles |
|---|---|---|
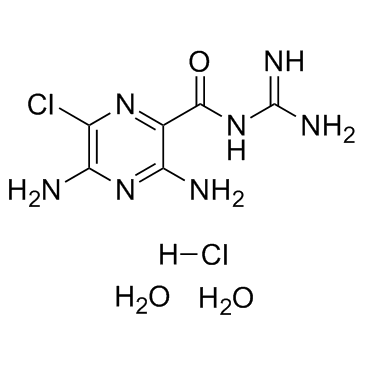 |
Amiloride HCl dihydrate
CAS:17440-83-4 |
|
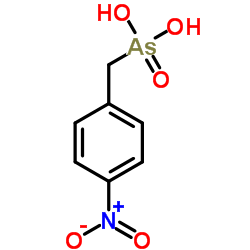 |
3′,5′-Cyclic nucleotide phosphodiesterase
CAS:9040-59-9 |