| Structure | Name/CAS No. | Articles |
|---|---|---|
 |
DL-Lysine
CAS:70-54-2 |
|
 |
Trichostatin A
CAS:58880-19-6 |
|
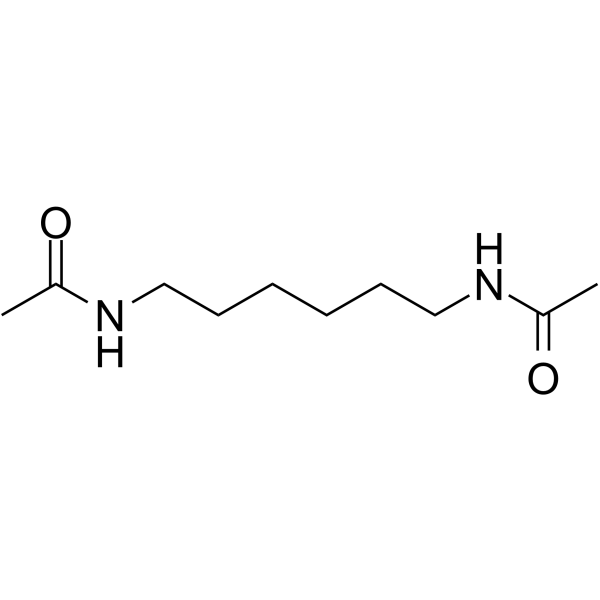 |
Hexamethylene bisacetamide
CAS:3073-59-4 |
|
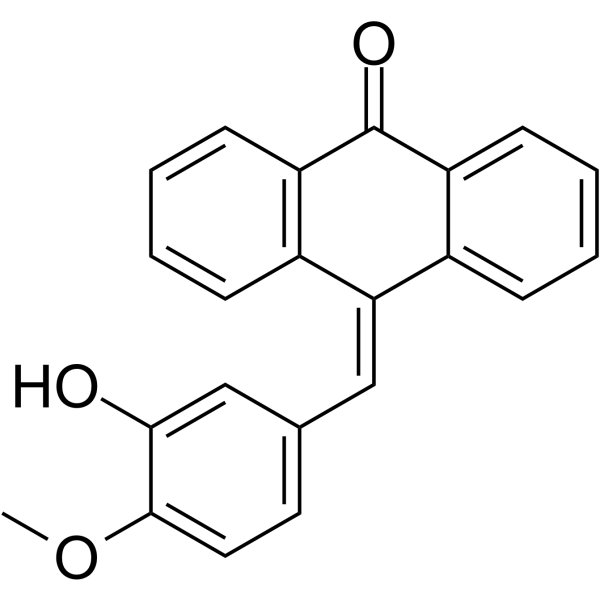 |
tubulin polymerization inhibitor
CAS:564468-51-5 |
|
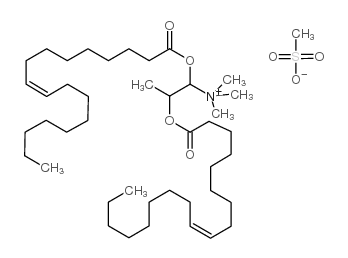 |
DOTAP Transfection Reagent
CAS:144189-73-1 |
|
 |
O-Phospho-L-serine
CAS:407-41-0 |