| Structure | Name/CAS No. | Articles |
|---|---|---|
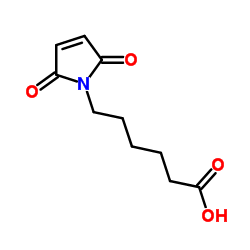 |
6-Maleimidocapronic acid
CAS:55750-53-3 |
|
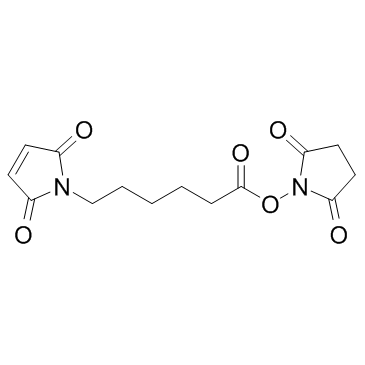 |
N-Succinimidyl 6-maleimidohexanoate
CAS:55750-63-5 |
|
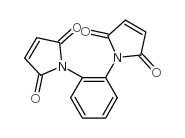 |
1H-Pyrrole-2,5-dione,1,1'-(1,2-phenylene)bis
CAS:13118-04-2 |
|
 |
2-iminothiolane
CAS:4781-83-3 |