| Structure | Name/CAS No. | Articles |
|---|---|---|
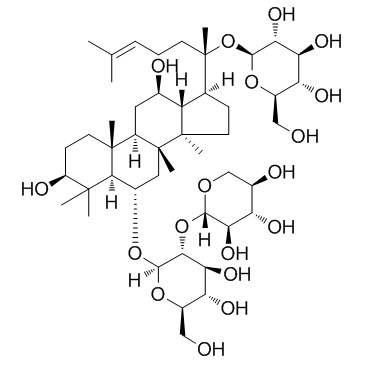 |
Notoginsenoside R1
CAS:80418-24-2 |
|
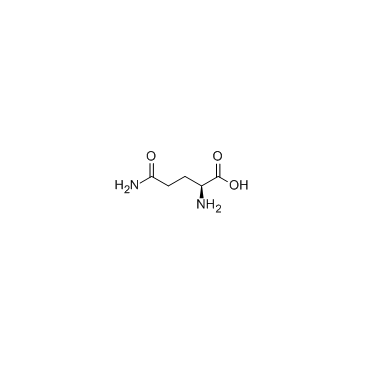 |
L-Glutamine
CAS:56-85-9 |
|
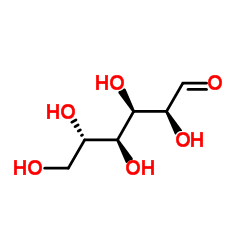 |
L-Glucose
CAS:921-60-8 |
|
 |
2,3,5-Triphenyltetrazolium chloride
CAS:298-96-4 |
|
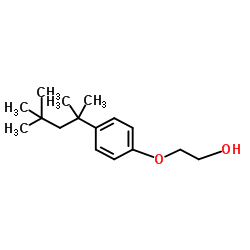 |
2-(4-(1,1,3,3-Tetramethylbutyl)phenoxy)ethanol
CAS:2315-67-5 |
|
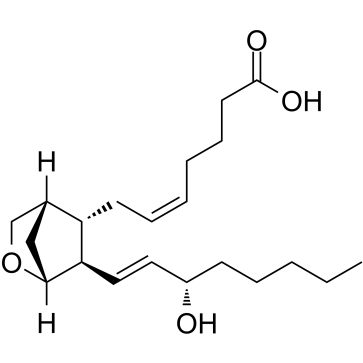 |
U-46619
CAS:56985-40-1 |
|
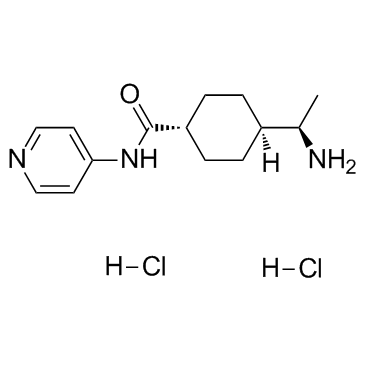 |
Y-27632 2HCl
CAS:129830-38-2 |