16941-11-0
| 中文名 | 六氟磷酸铵 |
|---|---|
| 英文名 | Ammonium hexafluorophosphate |
| 中文别名 | 六氟化酸铵 |
| 英文别名 |
Ammoniumhexafluorophosphate99.
NH4PF6,III EINECS 241-009-1 AMMONIUM HEXAFLUOPHOSPHATE Ammonium fluorophosphate Ammonium hexafluorophosphate MFCD00064642 Ammonium hexafluorop Phosphate(1-), hexafluoro-, ammonium (1:1) Azanium hexafluorophosphate |
| 密度 | 2.18 g/mL at 25 °C(lit.) |
|---|---|
| 熔点 | 198 °C |
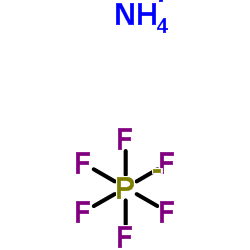
| 分子式 | H4F6NP |
| 分子量 | 163.00300 |
| 精确质量 | 162.99900 |
| PSA | 13.59000 |
| LogP | 3.75860 |
| 外观性状 | 白色结晶粉末 |
| 储存条件 | 本品应密封干燥保存。 |
| 稳定性 | 常温常压下稳定 避免的物料:水分/潮湿 酸 空气 热。易溶于水,可溶于丙酮、甲醇、乙醇和乙酸乙酯中。强烈加热时不经熔融即分解。与强酸共煮则慢慢水解。20℃时水中的溶解度为74.8g/100mL。 |
| 水溶解性 | 74.8 g/100 mL (20 ºC) |
| 计算化学 | 1.疏水参数计算参考值(XlogP):无 2.氢键供体数量:1 3.氢键受体数量:7 4.可旋转化学键数量:0 5.互变异构体数量:无 6.拓扑分子极性表面积1 7.重原子数量:8 8.表面电荷:0 9.复杂度:62.7 10.同位素原子数量:0 11.确定原子立构中心数量:0 12.不确定原子立构中心数量:0 13.确定化学键立构中心数量:0 14.不确定化学键立构中心数量:0 15.共价键单元数量:2 |
| 更多 | 1. 性状:无色正方叶状或薄片状结晶,很少有矩形板状的,等轴晶系。有吸湿性。在室温时不腐蚀玻璃。在热至相对高温时分解,并不预先熔化。 2. 密度(g/mL,25/4℃):2.180 3. 相对蒸汽密度(g/mL,空气=1):未确定 4. 熔点(ºC):未确定 5. 沸点(ºC,常压):未确定 6. 沸点(ºC,5.2kPa):未确定 7. 折射率:未确定 8. 闪点(ºC):未确定 9. 比旋光度(º):未确定 10. 自燃点或引燃温度(ºC):未确定 11. 蒸气压(kPa,25ºC):未确定 12. 饱和蒸气压(kPa,60ºC):未确定 13. 燃烧热(KJ/mol):未确定 14. 临界温度(ºC):未确定 15. 临界压力(KPa):未确定 16. 油水(辛醇/水)分配系数的对数值:未确定 17. 爆炸上限(%,V/V):未确定 18. 爆炸下限(%,V/V):未确定 19. 溶解性:水中溶解度(20℃)74.8g/100ml。溶于丙酮、甲醇、乙醇、乙酸甲酯。在沸强酸中缓慢水解。 |
Synonym:None Section 2 - COMPOSITION, INFORMATION ON INGREDIENTS
Risk Phrases: 34 Section 3 - HAZARDS IDENTIFICATION EMERGENCY OVERVIEW
Causes burns.Hygroscopic (absorbs moisture from the air).Corrosive. Potential Health Effects Eye: Causes eye burns. Skin: Causes skin burns. Ingestion: May cause severe and permanent damage to the digestive tract. Causes gastrointestinal tract burns. Inhalation: May cause severe irritation of the respiratory tract with sore throat, coughing, shortness of breath and delayed lung edema. Causes chemical burns to the respiratory tract. Chronic: No information found. Section 4 - FIRST AID MEASURES Eyes: Get medical aid immediately. Do NOT allow victim to rub eyes or keep eyes closed. Extensive irrigation with water is required (at least 30 minutes). Skin: Get medical aid immediately. Immediately flush skin with plenty of water for at least 15 minutes while removing contaminated clothing and shoes. Wash clothing before reuse. Destroy contaminated shoes. Ingestion: Do not induce vomiting. If victim is conscious and alert, give 2-4 cupfuls of milk or water. Never give anything by mouth to an unconscious person. Get medical aid immediately. Inhalation: Get medical aid immediately. Remove from exposure and move to fresh air immediately. If not breathing, give artificial respiration. If breathing is difficult, give oxygen. Do NOT use mouth-to-mouth resuscitation. Notes to Physician: Section 5 - FIRE FIGHTING MEASURES General Information: As in any fire, wear a self-contained breathing apparatus in pressure-demand, MSHA/NIOSH (approved or equivalent), and full protective gear. During a fire, irritating and highly toxic gases may be generated by thermal decomposition or combustion. Vapors may be heavier than air. They can spread along the ground and collect in low or confined areas. Extinguishing Media: In case of fire, use water, dry chemical, chemical foam, or alcohol-resistant foam. Do NOT get water inside containers. Section 6 - ACCIDENTAL RELEASE MEASURES General Information: Use proper personal protective equipment as indicated in Section 8. Spills/Leaks: Clean up spills immediately, observing precautions in the Protective Equipment section. Sweep up, then place into a suitable container for disposal. Avoid generating dusty conditions. Provide ventilation. Do not get water inside containers. Section 7 - HANDLING and STORAGE Handling: Wash hands before eating. Keep container tightly closed. Do not get on skin or in eyes. Do not ingest or inhale. Use with adequate ventilation. Wash clothing before reuse. Discard contaminated shoes. Keep from contact with moist air and steam. Storage: Keep container closed when not in use. Store in a cool, dry, well-ventilated area away from incompatible substances. Corrosives area. Section 8 - EXPOSURE CONTROLS, PERSONAL PROTECTION Engineering Controls: Facilities storing or utilizing this material should be equipped with an eyewash facility and a safety shower. Use adequate ventilation to keep airborne concentrations low. Exposure Limits CAS# 16941-11-0: Personal Protective Equipment Eyes: Wear appropriate protective eyeglasses or chemical safety goggles as described by OSHA's eye and face protection regulations in 29 CFR 1910.133 or European Standard EN166. Skin: Wear appropriate protective gloves to prevent skin exposure. Clothing: Wear appropriate protective clothing to minimize contact with skin. Respirators: A respiratory protection program that meets OSHA's 29 CFR 1910.134 and ANSI Z88.2 requirements or European Standard EN 149 must be followed whenever workplace conditions warrant respirator use. Section 9 - PHYSICAL AND CHEMICAL PROPERTIES Physical State: Crystalline powder Color: white Odor: None reported. pH: Not available. Vapor Pressure: Not available. Viscosity: Not available. Boiling Point: Not available. Freezing/Melting Point: 198 deg C Autoignition Temperature: Not available. Flash Point: Not available. Explosion Limits, lower: N/A Explosion Limits, upper: N/A Decomposition Temperature: Solubility in water: 74.8 g/100ml (20 c) Specific Gravity/Density: 2.1800g/cm3 Molecular Formula: H4F6NP Molecular Weight: 163.00 Section 10 - STABILITY AND REACTIVITY Chemical Stability: Stable under normal temperatures and pressures. Conditions to Avoid: Incompatible materials, dust generation, excess heat, oxidizers. Incompatibilities with Other Materials: Oxidizing agents. Hazardous Decomposition Products: Phosphine, oxides of phosphorus, hydrogen fluoride gas, ammonia and/or derivatives. Hazardous Polymerization: Has not been reported. Section 11 - TOXICOLOGICAL INFORMATION RTECS#: CAS# 16941-11-0 unlisted. LD50/LC50: Not available. Carcinogenicity: Ammonium Hexafluorophosphate - Not listed by ACGIH, IARC, or NTP. Section 12 - ECOLOGICAL INFORMATION Section 13 - DISPOSAL CONSIDERATIONS Dispose of in a manner consistent with federal, state, and local regulations. Section 14 - TRANSPORT INFORMATION IATA Shipping Name: CORROSIVE SOLID, ACIDIC, INORGANIC, N.O.S.* Hazard Class: 8 UN Number: 3260 Packing Group: II IMO Shipping Name: CORROSIVE SOLID, ACIDIC, INORGANIC, N.O.S. Hazard Class: 8 UN Number: 3260 Packing Group: II RID/ADR Shipping Name: CORROSIVE SOLID, ACIDIC, INORGANIC, N.O.S. Hazard Class: 8 UN Number: 3260 Packing group: II Section 15 - REGULATORY INFORMATION European/International Regulations European Labeling in Accordance with EC Directives Hazard Symbols: C Risk Phrases: R 34 Causes burns. Safety Phrases: S 26 In case of contact with eyes, rinse immediately with plenty of water and seek medical advice. WGK (Water Danger/Protection) CAS# 16941-11-0: No information available. Canada CAS# 16941-11-0 is listed on Canada's NDSL List. CAS# 16941-11-0 is not listed on Canada's Ingredient Disclosure List. US FEDERAL TSCA CAS# 16941-11-0 is listed on the TSCA inventory. SECTION 16 - ADDITIONAL INFORMATION N/A |
|
生态学数据: 通常对水体是稍微有害的,不要将未稀释或大量产品接触地下水,水道或污水系统,未经政府许可勿将材料排入周围环境。
|
| 符号 |

GHS05 |
|---|---|
| 信号词 | Danger |
| 危害声明 | H314 |
| 警示性声明 | P280-P305 + P351 + P338-P310 |
| 个人防护装备 | Eyeshields;Faceshields;full-face particle respirator type N100 (US);Gloves;respirator cartridge type N100 (US);type P1 (EN143) respirator filter;type P3 (EN 143) respirator cartridges |
| 危害码 (欧洲) | C:Corrosive; |
| 风险声明 (欧洲) | R34 |
| 安全声明 (欧洲) | S26-S36/37/39-S45 |
| 危险品运输编码 | UN 3260 8/PG 2 |
| WGK德国 | 3 |
| 包装等级 | III |
| 危险类别 | 8 |