| 结构式 | 名称/CAS号 | 全部文献 |
|---|---|---|
 |
CK-666
CAS:442633-00-3 |
|
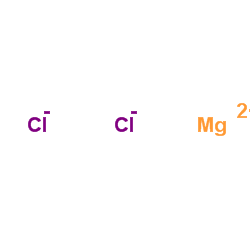 |
氯化镁
CAS:7786-30-3 |
|
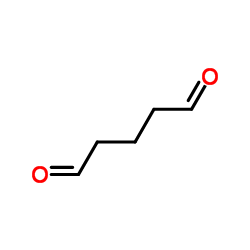 |
戊二醛
CAS:111-30-8 |
|
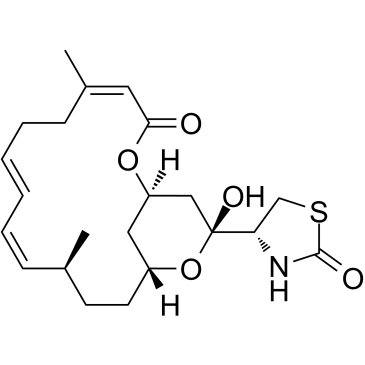 |
红海海绵素 A
CAS:76343-93-6 |
|
 |
吗啉乙磺酸
CAS:4432-31-9 |