| 结构式 | 名称/CAS号 | 全部文献 |
|---|---|---|
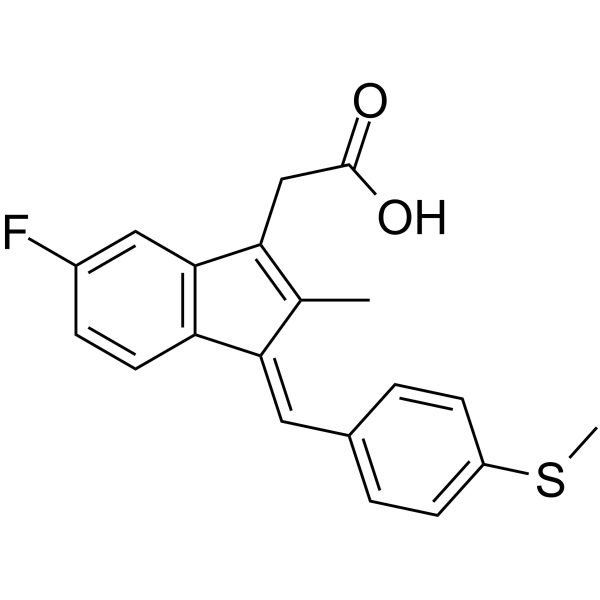 |
5-氟-2-甲基-1-(4-甲硫基亚苄基)茚-3-乙酸
CAS:32004-67-4 |
|
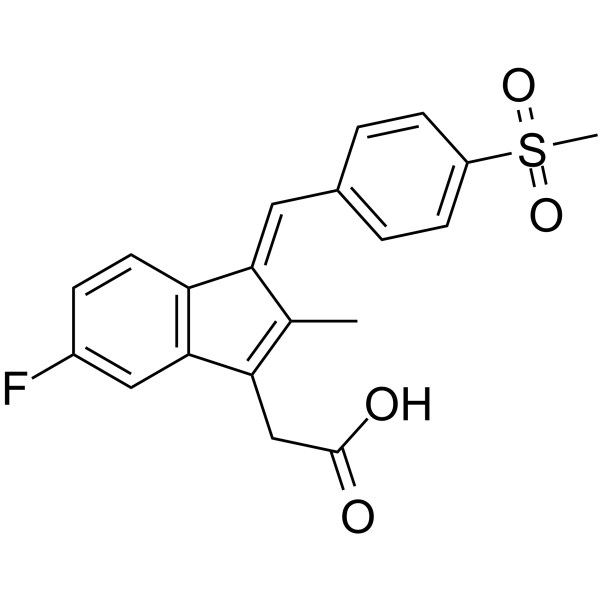 |
Sulindac sulfone
CAS:59864-04-9 |