| 结构式 | 名称/CAS号 | 全部文献 |
|---|---|---|
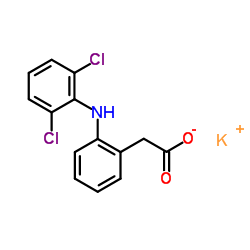 |
双氯芬酸钾
CAS:15307-81-0 |
|
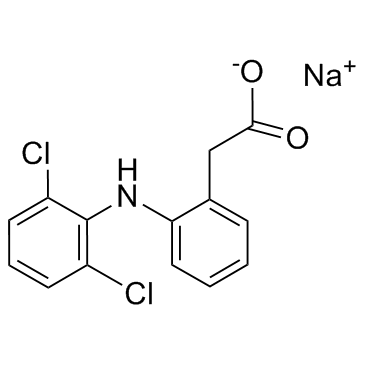 |
双氯芬酸钠
CAS:15307-79-6 |
|
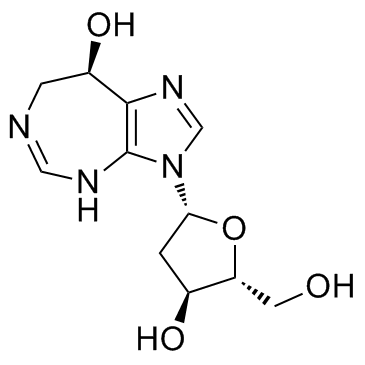 |
喷司他丁
CAS:53910-25-1 |