| Structure | Name/CAS No. | Articles |
|---|---|---|
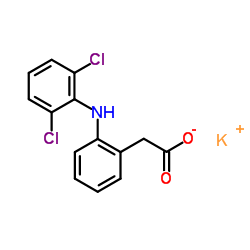 |
Diclofenac potassium
CAS:15307-81-0 |
|
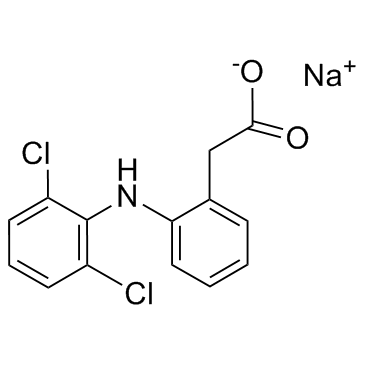 |
Diclofenac sodium
CAS:15307-79-6 |
|
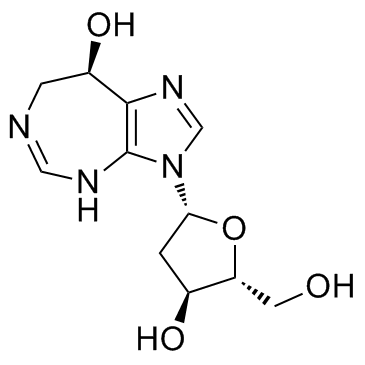 |
PENTOSTATIN
CAS:53910-25-1 |