Toxoflavin
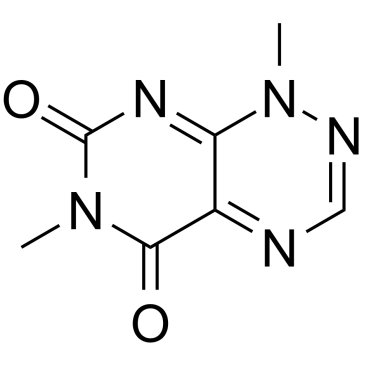
Toxoflavin structure
|
Common Name | Toxoflavin | ||
|---|---|---|---|---|
| CAS Number | 84-82-2 | Molecular Weight | 193.16300 | |
| Density | 1.65g/cm3 | Boiling Point | 276.7ºC at 760 mmHg | |
| Molecular Formula | C7H7N5O2 | Melting Point | N/A | |
| MSDS | Chinese USA | Flash Point | 121.1ºC | |
| Symbol |

GHS06 |
Signal Word | Danger | |
Use of ToxoflavinToxoflavin (Xanthothricin) is an antagonist of transcription factor 4 (TCF4)/β-catenin complex, also acts as an inhibitor of KDM4A, with antitumor activity[1][2]. |
| Name | 1,6-dimethylpyrimido[5,4-e][1,2,4]triazine-5,7-dione |
|---|---|
| Synonym | More Synonyms |
| Description | Toxoflavin (Xanthothricin) is an antagonist of transcription factor 4 (TCF4)/β-catenin complex, also acts as an inhibitor of KDM4A, with antitumor activity[1][2]. |
|---|---|
| Related Catalog | |
| Target |
TCF4/β-catenin[1], KDM4A[2] |
| In Vitro | Toxoflavin (Xanthothricin) exhibits dose-dependent cytotoxicity against the 3 hepatoma cell lines, and with IC50s of 0.66 μM, 0.36 μM and 0.98 μM for Hep40 cells, HepG2 cells and Huh7 cells respectively[1]. |
| References |
| Density | 1.65g/cm3 |
|---|---|
| Boiling Point | 276.7ºC at 760 mmHg |
| Molecular Formula | C7H7N5O2 |
| Molecular Weight | 193.16300 |
| Flash Point | 121.1ºC |
| Exact Mass | 193.06000 |
| PSA | 82.67000 |
| Index of Refraction | 1.76 |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Symbol |

GHS06 |
|---|---|
| Signal Word | Danger |
| Hazard Statements | H300 |
| Precautionary Statements | P264-P301 + P310 |
| RIDADR | UN 2811 6.1 / PGIII |
| HS Code | 2933990090 |
| HS Code | 2933990090 |
|---|---|
| Summary | 2933990090. heterocyclic compounds with nitrogen hetero-atom(s) only. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
|
Investigations into the Biosynthesis, Regulation, and Self-Resistance of Toxoflavin in Pseudomonas protegens Pf-5.
ChemBioChem. 16 , 1782-90, (2015) Pseudomonas spp. are prolific producers of natural products from many structural classes. Here we show that the soil bacterium Pseudomonas protegens Pf-5 is capable of producing trace levels of the tr... |
|
|
The quorum sensing-dependent gene katG of Burkholderia glumae is important for protection from visible light.
J. Bacteriol. 191(13) , 4152-7, (2009) Quorum sensing (QS) plays important roles in the pathogenicity of Burkholderia glumae, the causative agent of bacterial rice grain rot. We determined how QS is involved in catalase expression in B. gl... |
|
|
Identification of the product of toxoflavin lyase: degradation via a Baeyer-Villiger oxidation.
J. Am. Chem. Soc. 134(11) , 5326-30, (2012) Toxoflavin (an azapteridine) is degraded to a single product by toxoflavin lyase (TflA) in a reaction dependent on reductant, Mn(II), and oxygen. The isolated product was fully characterized by NMR an... |
| Toxoflavin |
| 1,6-Dimethyl-1H-pyrimido[5,4-e][1,2,4]triazin-5,7-dion |
| 1,6-dimethylpyrimido[5,4-e]-1,2,4-triazine-5,7(1H,6H)-dione |
| Xanthotricin |
| Xanthothricin (VAN) |
| Compound 7215 |
| Toxoflavine |
| Xanthothricin |
| 1,6-dimethyl-1H-pyrimido[5,4-e][1,2,4]triazine-5,7-dione |
| GNF-Pf-67 |