Doxazosin mesylate
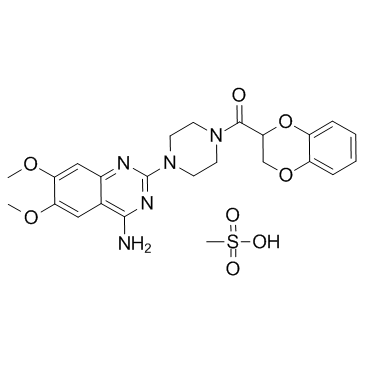
Doxazosin mesylate structure
|
Common Name | Doxazosin mesylate | ||
|---|---|---|---|---|
| CAS Number | 77883-43-3 | Molecular Weight | 547.581 | |
| Density | N/A | Boiling Point | 718ºC at 760 mmHg | |
| Molecular Formula | C24H29N5O8S | Melting Point | 275-277ºC | |
| MSDS | Chinese USA | Flash Point | 388ºC | |
Use of Doxazosin mesylateDoxazosin mesylate(UK 33274) is a quinazoline-derivative that selectively antagonizes postsynaptic α1-adrenergic receptors.Target: α1-adrenergic receptorDoxazosin (mesylate) is the mesylate salt form of doxazosin, which is a long-lasting inhibitor of α1-adrenoceptors that is widely used to treat benign prostatic hyperplasia and lower urinary tract symptoms [1]. doxazosin may have a direct inhibitory effect on cholesterol synthesis independent of the LDL receptor. The inhibition of cholesterol synthesis by doxazosin may cause cells to compensate by upregulating the LDL receptor, thereby increasing the importation of lipoprotein cholesterol and reducing LDL cholesterol in the medium [2]. Doxazosin monotherapy was effective in eight of 12 patients (66.7%), and combined therapy with a beta-blocker was effective in 11 of 12 patients (91.7%). The mean pulse rate remained constant throughout therapy. Adverse reactions were minor and transient and occurred in only three patients. Urinary and plasma catecholamine levels tended to decrease or remained unchanged during doxazosin therapy [3]. |
| Name | doxazosin mesylate |
|---|---|
| Synonym | More Synonyms |
| Description | Doxazosin mesylate(UK 33274) is a quinazoline-derivative that selectively antagonizes postsynaptic α1-adrenergic receptors.Target: α1-adrenergic receptorDoxazosin (mesylate) is the mesylate salt form of doxazosin, which is a long-lasting inhibitor of α1-adrenoceptors that is widely used to treat benign prostatic hyperplasia and lower urinary tract symptoms [1]. doxazosin may have a direct inhibitory effect on cholesterol synthesis independent of the LDL receptor. The inhibition of cholesterol synthesis by doxazosin may cause cells to compensate by upregulating the LDL receptor, thereby increasing the importation of lipoprotein cholesterol and reducing LDL cholesterol in the medium [2]. Doxazosin monotherapy was effective in eight of 12 patients (66.7%), and combined therapy with a beta-blocker was effective in 11 of 12 patients (91.7%). The mean pulse rate remained constant throughout therapy. Adverse reactions were minor and transient and occurred in only three patients. Urinary and plasma catecholamine levels tended to decrease or remained unchanged during doxazosin therapy [3]. |
|---|---|
| Related Catalog | |
| References |
| Boiling Point | 718ºC at 760 mmHg |
|---|---|
| Melting Point | 275-277ºC |
| Molecular Formula | C24H29N5O8S |
| Molecular Weight | 547.581 |
| Flash Point | 388ºC |
| Exact Mass | 547.173706 |
| PSA | 175.02000 |
| LogP | 2.88670 |
| Storage condition | Desiccate at RT |
| Stability | Protect from light |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
|
Systems pharmacology identifies drug targets for Stargardt disease-associated retinal degeneration.
J. Clin. Invest. 123(12) , 5119-34, (2013) A systems pharmacological approach that capitalizes on the characterization of intracellular signaling networks can transform our understanding of human diseases and lead to therapy development. Here,... |
|
|
The efficacy and safety of alpha-1 blockers for benign prostatic hyperplasia: an overview of 15 systematic reviews.
Curr. Med. Res. Opin. 29(3) , 279-87, (2013) A great number of clinical trials and systematic reviews have evaluated the efficacy and safety of α(1) blockers for benign prostatic hyperplasia (BPH). We carried out an overview of reviews to provid... |
|
|
[Results of open multicenter study of the safety of doxazosin in combination with indigal in men with stages I-II prostatic adenoma].
Urologiia. (2) , 42-4, 46, (2013) The article presents a method of conservative treatment of men with I-II stage prostatic adenoma using a combination of doxazosin and indigal, which has antioxidant, antiproliferative and anti-inflamm... |
| piperazine, 1-(4-amino-6,7-dimethoxy-2-quinazolinyl)-4-[(2,3-dihydro-1,4-benzodioxin-2-yl)carbonyl]-, methanesulfonate (1:1) |
| Normothen |
| [4-(4-Amino-6,7-dimethoxyquinazolin-2-yl)piperazin-1-yl](2,3-dihydro-1,4-benzodioxin-2-yl)methanone methanesulfonate |
| [4-(4-Amino-6,7-dimethoxyquinazolin-2-yl)piperazin-1-yl](2,3-dihydro-1,4-benzodioxin-2-yl)methanone methanesulfonate (1:1) |
| Doxazosin mesylate |
| Hydrogen methanesulfonate - [4-(4-amino-6,7-dimethoxy-2-quinazolinyl)-1-piperazinyl](2,3-dihydro-1,4-benzodioxin-2-yl)methanone (1:1:1) |
| Cardura |
| MFCD00216023 |
| Methanone, [4-(4-amino-6,7-dimethoxy-2-quinazolinyl)-1-piperazinyl](2,3-dihydro-1,4-benzodioxin-2-yl)-, methanesulfonate (1:1) |
| UNII:86P6PQK0MU |
| [4-(4-Amino-6,7-dimethoxy-2-quinazolinyl)-1-piperazinyl](2,3-dihydro-1,4-benzodioxin-2-yl)methanone methanesulfonate |
| [4-(4-amino-6,7-dimethoxyquinazolin-2-yl)piperazin-1-yl]-(2,3-dihydro-1,4-benzodioxin-3-yl)methanone,methanesulfonic acid |
| Doxazosin (mesylate) |

