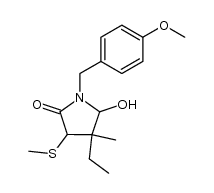
Ethosuximide

Ethosuximide structure
|
Common Name | Ethosuximide | ||
|---|---|---|---|---|
| CAS Number | 77-67-8 | Molecular Weight | 141.168 | |
| Density | 1.1±0.1 g/cm3 | Boiling Point | 265.3±9.0 °C at 760 mmHg | |
| Molecular Formula | C7H11NO2 | Melting Point | 51ºC | |
| MSDS | Chinese USA | Flash Point | 123.8±18.9 °C | |
| Symbol |

GHS07 |
Signal Word | Warning | |
Use of EthosuximideEthosuximide, a widely prescribed anti-epileptic drug, improves the phenotypes of multiple neurodegenerative disease models and blocks the low voltage activated T-type calcium channel. |
| Name | ethosuximide |
|---|---|
| Synonym | More Synonyms |
| Description | Ethosuximide, a widely prescribed anti-epileptic drug, improves the phenotypes of multiple neurodegenerative disease models and blocks the low voltage activated T-type calcium channel. |
|---|---|
| Related Catalog | |
| Target |
calcium channel[1] |
| In Vitro | The efficacy of Ethosuximide in generalized absence epilepsy is thought to be due to blockade of the low voltage activated T-type calcium channel. There is no reduction in total Tau levels in Ethosuximide treated Tau transgenic worms as compare to vehicle controls. The rescuing effect of Ethosuximide is therefore not due to transgene suppression or reduced expression of toxic mutant Tau protein. Quantification of the amount of soluble and insoluble (RIPA-extractable) Tau relative to total Tau levels reveals a significant reduction in aberrantly-folded, insoluble Tau and a corresponding increase in soluble Tau in Ethosuximide-treated compare with untreated worms[1]. Concentrations of 2 μM or more of Ethosuximide not only are found to be less effective than 1 μM concentration of Ethosuximide, but also induce cell toxicity. GABA staining immunofluorescence images show that after treatment with Ethosuximide, GABA positive neuron increases by 3 and 6.5 fold for concentrations of 0.1 and 1 μM, respectively. BrdU staining shows nuclei proliferation after 2 to 3 days of Ethosuximide exposure. The mean of nuclei is 15.98±0.41 for the low concentration of Ethosuximide while it is 25.27±0.48 for the high concentration after Brdu staining. This number is 11.05±0.2 for lithium chloride[2]. |
| Kinase Assay | Vehicle- and Ethosuximide-treated Tau V337M worms are lysed and separated into soluble and insoluble fractions. Fractions are separated by SDS-PAGE and western blotted using anti-human Tau T46 and anti-actin antibodies. The abundance of Tau protein in each fraction is quantified by densitometry and normalized against beta-actin. Total Tau levels in lysates are expressed as the percentage of actin-normalized Tau relative to vehicle control lysates; Tau levels in sequentially extracted fractions are expressed as the percentage of actin-normalized Tau relative to the sum of both fractions (soluble+RIPA) combined[1]. |
| Cell Assay | Neuronal stem cells from the forebrain Cortex of a 3-day-old rat are used in this study. The cells are differentiated by withdrawal of basic fibroblastic growth factor (bFGF) and exposed to Ethosuximide at two concentrations of 0.1 μM and 1 μM. Before drug treatment, the cells are rinsed once with PBS, and the medium is replaced with fresh, bFGF-free DMEM/F12 medium containing different concentration of Ethosuximide. Medium exchange is done every day for 6 days with medium containing Ethosuximide. Then, cells are fixed for immunocytochemistry[2]. |
| References |
| Density | 1.1±0.1 g/cm3 |
|---|---|
| Boiling Point | 265.3±9.0 °C at 760 mmHg |
| Melting Point | 51ºC |
| Molecular Formula | C7H11NO2 |
| Molecular Weight | 141.168 |
| Flash Point | 123.8±18.9 °C |
| Exact Mass | 141.078979 |
| PSA | 46.17000 |
| LogP | 0.38 |
| Vapour Pressure | 0.0±0.5 mmHg at 25°C |
| Index of Refraction | 1.451 |
| Storage condition | Refrigerator |
| Water Solubility | ethanol: 100 mg/mL |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
MUTATION DATA
|
| Symbol |

GHS07 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H302 |
| Precautionary Statements | P301 + P312 + P330 |
| Personal Protective Equipment | dust mask type N95 (US);Eyeshields;Gloves |
| Hazard Codes | Xn: Harmful; |
| Risk Phrases | R22 |
| Safety Phrases | 36 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| RTECS | WN2800000 |
| HS Code | 2925190090 |
|
~% 
Ethosuximide CAS#:77-67-8 |
| Literature: Journal of Organic Chemistry, , vol. 46, p. 2757 |
|
~47% 
Ethosuximide CAS#:77-67-8 |
| Literature: Ishibashi, Hiroyuki; Nakaharu, Tohru; Nishimura, Masako; Nishikawa, Atsuko; Kameoka, Chisato; Ikeda, Masazumi Tetrahedron, 1995 , vol. 51, # 10 p. 2929 - 2938 |
|
~% 
Ethosuximide CAS#:77-67-8 |
| Literature: Tetrahedron, , vol. 51, # 10 p. 2929 - 2938 |
|
~% 
Ethosuximide CAS#:77-67-8 |
| Literature: Tetrahedron, , vol. 51, # 10 p. 2929 - 2938 |
|
~% 
Ethosuximide CAS#:77-67-8 |
| Literature: Tetrahedron, , vol. 51, # 10 p. 2929 - 2938 |
|
~% 
Ethosuximide CAS#:77-67-8 |
| Literature: Tetrahedron, , vol. 51, # 10 p. 2929 - 2938 |
| HS Code | 2925190090 |
|---|---|
| Summary | 2925190090 other imides and their derivatives; salts thereof VAT:17.0% Tax rebate rate:9.0% Supervision conditions:none MFN tariff:6.5% General tariff:30.0% |
|
Ethosuximide and phenytoin dose-dependently attenuate acute nonconvulsive seizures after traumatic brain injury in rats.
J. Neurotrauma 30(23) , 1973-82, (2013) Acute seizures frequently occur following severe traumatic brain injury (TBI) and have been associated with poor patient prognosis. Silent or nonconvulsive seizures (NCS) manifest in the absence of mo... |
|
|
How did phenobarbital's chemical structure affect the development of subsequent antiepileptic drugs (AEDs)?
Epilepsia 53 Suppl 8 , 3-11, (2012) Phenobarbital has been in clinical use as an antiepileptic drug (AED) since 1912. The initial clinical success of phenobarbital and other barbiturates affected the design of subsequent AEDs (e.g., phe... |
|
|
Histone deacetylase inhibitor valproic acid promotes the differentiation of human induced pluripotent stem cells into hepatocyte-like cells.
PLoS ONE 9(8) , e104010, (2014) In this study, we aimed to elucidate the effects and mechanism of action of valproic acid on hepatic differentiation from human induced pluripotent stem cell-derived hepatic progenitor cells. Human in... |
| (±)-Ethosuximide |
| 2-Methyl-2-ethylsuccinimide |
| 3-Ethyl-3-methylpyrrolidine-2,5-dione |
| 2-Ethyl-2-methylsuccinimide |
| MFCD00072123 |
| Ethymal |
| Zarontin |
| α-Ethyl-α-methylsuccinimide |
| Etosuximida |
| 2,5-Pyrrolidinedione, 3-ethyl-3-methyl- |
| Zarondan |
| Petinimid |
| Suxilep |
| 3-ethyl-3-methyl-pyrrolidine-2,5-dione |
| Ethosuximide |
| 2,5-Pyrrolidinedione, 3-ethyl-3-methyl-, (±)- |
| Emeside |
| thosuximide |
| Suxinutin |
| EINECS 201-048-7 |
| UNII:5SEH9X1D1D |
| 3-ethyl-3-methylsuccinimide |
| (±)-2-Ethyl-2-methylsuccinimide |
| 3-Aethyl-3-methyl-pyrrolidin-2,5-dion |
| Petnidan |
| 3-Ethyl-3-methyl-2,5-pyrrolidinedione |