2-Methyl-4-nitroimidazole
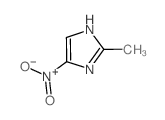
2-Methyl-4-nitroimidazole structure
|
Common Name | 2-Methyl-4-nitroimidazole | ||
|---|---|---|---|---|
| CAS Number | 696-23-1 | Molecular Weight | 127.10100 | |
| Density | 1.426g/cm3 | Boiling Point | 399.5ºC at 760 mmHg | |
| Molecular Formula | C4H5N3O2 | Melting Point | 251-255ºC(lit.) | |
| MSDS | Chinese USA | Flash Point | 195.4ºC | |
| Symbol |

GHS08 |
Signal Word | Warning | |
| Name | 2-Methyl-4(5)-nitroimidazole |
|---|---|
| Synonym | More Synonyms |
| Density | 1.426g/cm3 |
|---|---|
| Boiling Point | 399.5ºC at 760 mmHg |
| Melting Point | 251-255ºC(lit.) |
| Molecular Formula | C4H5N3O2 |
| Molecular Weight | 127.10100 |
| Flash Point | 195.4ºC |
| Exact Mass | 127.03800 |
| PSA | 74.50000 |
| LogP | 1.14950 |
| Index of Refraction | 1.591 |
| Storage condition | Refrigerator |
| Stability | Stable. Incompatible with strong oxidizing agents. |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATAMUTATION DATA
|
| Symbol |

GHS08 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H351 |
| Precautionary Statements | P281 |
| Personal Protective Equipment | dust mask type N95 (US);Eyeshields;Gloves |
| Hazard Codes | Xn |
| Risk Phrases | R22 |
| Safety Phrases | S22-S36/37 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| RTECS | NI7550000 |
| HS Code | 2933290090 |
|
~89% 
2-Methyl-4-nitr... CAS#:696-23-1 |
| Literature: BASF Aktiengesellschaft Patent: US4145553 A1, 1979 ; |
|
~86% 
2-Methyl-4-nitr... CAS#:696-23-1 |
| Literature: Journal of Chemical Research - Part S, , # 5 p. 244 - 245 |
|
~22% 
2-Methyl-4-nitr... CAS#:696-23-1 |
| Literature: Polish Journal of Chemistry, , vol. 61, # 7-12 p. 913 - 920 |
|
~% 
2-Methyl-4-nitr... CAS#:696-23-1 |
| Literature: Polish Journal of Chemistry, , vol. 66, # 5 p. 819 - 826 |
| Precursor 1 | |
|---|---|
| DownStream 9 | |
| HS Code | 2933290090 |
|---|---|
| Summary | 2933290090. other compounds containing an unfused imidazole ring (whether or not hydrogenated) in the structure. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
|
On the hydrolytic behavior of tinidazole, metronidazole, and ornidazole.
J. Pharm. Sci. 92(4) , 739-46, (2003) Using two UV-spectrophotometric methods, the hydrolysis of tinidazole was studied at pH 1.00-8.45 at 80 degrees C. The reaction followed apparent first-order kinetics throughout the studied range. No ... |
|
|
Radicals of nitroimidazole derivatives: pH dependence of rates of formation and decay related to acid-base equilibria.
Int. J. Radiat. Biol. Relat. Stud. Phys. Chem. Med. 51(5) , 797-809, (1987) Three analogous 5-nitroimidazoles, having radiosensitizing and cytotoxic properties, have been studied by pulse-radiolysis in N2O-saturated aqueous formate solutions. Rates of formation of the radical... |
|
|
[Synthesis and antibacterial activity of ciprofloxacin derivatives].
Yao Xue Xue Bao 40(2) , 132-5, (2005) To study the synthesis and antibacterial activity of ciprofloxacin derivatives.Ciprofloxacin derivatives were synthesized primarily from 2-methyl-5-nitroimidazol and ciprofloxacin through nucleophilic... |
| 2-Methyl-4-nitro-1H-imidazole |
| EINECS 211-790-3 |
| MFCD00005191 |
| 2-Methyl-4-nitroimidazole |
| Metronidazole Impurity 1 |
![2-methyl-4-nitro-1-[(4-nitrophenyl)methyl]imidazole structure](https://image.chemsrc.com/caspic/378/107108-25-8.png) CAS#:107108-25-8
CAS#:107108-25-8 CAS#:13230-04-1
CAS#:13230-04-1 CAS#:6058-57-7
CAS#:6058-57-7 CAS#:14419-11-5
CAS#:14419-11-5 CAS#:443-48-1
CAS#:443-48-1 CAS#:3964-18-9
CAS#:3964-18-9 CAS#:117836-32-5
CAS#:117836-32-5
