4-Amino-1-benzylpiperidine

4-Amino-1-benzylpiperidine structure
|
Common Name | 4-Amino-1-benzylpiperidine | ||
|---|---|---|---|---|
| CAS Number | 50541-93-0 | Molecular Weight | 190.285 | |
| Density | 1.0±0.1 g/cm3 | Boiling Point | 281.2±33.0 °C at 760 mmHg | |
| Molecular Formula | C12H18N2 | Melting Point | 90-93ºC | |
| MSDS | Chinese USA | Flash Point | 113.9±20.5 °C | |
| Symbol |

GHS07 |
Signal Word | Warning | |
| Name | 4-Amino-1-benzylpiperidine |
|---|---|
| Synonym | More Synonyms |
| Density | 1.0±0.1 g/cm3 |
|---|---|
| Boiling Point | 281.2±33.0 °C at 760 mmHg |
| Melting Point | 90-93ºC |
| Molecular Formula | C12H18N2 |
| Molecular Weight | 190.285 |
| Flash Point | 113.9±20.5 °C |
| Exact Mass | 190.147003 |
| PSA | 29.26000 |
| LogP | 1.36 |
| Vapour Pressure | 0.0±0.6 mmHg at 25°C |
| Index of Refraction | 1.561 |
| Storage condition | 2-8°C |
| Water Solubility | immiscible |
| Symbol |

GHS07 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H315-H319-H335 |
| Precautionary Statements | P261-P305 + P351 + P338 |
| Personal Protective Equipment | Eyeshields;full-face respirator (US);Gloves;multi-purpose combination respirator cartridge (US);type ABEK (EN14387) respirator filter |
| Hazard Codes | Xi:Irritant; |
| Risk Phrases | R36/37/38 |
| Safety Phrases | S26-S36 |
| RIDADR | NONH for all modes of transport |
| WGK Germany | 3 |
| HS Code | 29333999 |
| Precursor 10 | |
|---|---|
| DownStream 10 | |
| HS Code | 2933399090 |
|---|---|
| Summary | 2933399090. other compounds containing an unfused pyridine ring (whether or not hydrogenated) in the structure. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:6.5%. General tariff:20.0% |
|
Studies on the collision-induced dissociation of adipoR agonists after electrospray ionization and their implementation in sports drug testing.
J. Mass Spectrom. 50(2) , 407-17, (2015) AdipoR agonists are small, orally active molecules capable of mimicking the protein adiponectin, which represents an adipokine with antidiabetic and antiatherogenic effects. Two adiponectin receptors ... |
|
|
N-Benzylpiperidine derivatives of 1,2,4-thiadiazolidinone as new acetylcholinesterase inhibitors.
Eur. J. Med. Chem. 35(10) , 913-22, (2000) A new family of 1,2,4-thiadiazolidinone derivatives containing the N-benzylpiperidine fragment has been synthesised. The acetylcholinesterase (AChE) inhibitory activity of all compounds was measured u... |
|
|
Synthesis of new glycyrrhetinic acid (GA) derivatives and their effects on tyrosinase activity.
Bioorg. Med. Chem. 11(24) , 5345-52, (2003) To synthesize glycyrrhetinic acid (GA) derivatives (3, 4, 5, 10, 13, 14, 15, and 16), we first removed the ketonic group in the C-11 position, and the carboxylic function at the C-30 position was kept... |
| 4-Piperidin-1-ylmethyl-phenylamine |
| 4-amino-benzylpiperidine |
| MFCD03422516 |
| N-Benzyl-4-amino piperidine |
| 4-(Piperidin-1-ylmethyl)benzenamine |
| 4-Piperidinamine, 1-(phenylmethyl)- |
| 1-benzyl-4-aMino |
| EINECS 256-620-9 |
| 4-Amino-1-Benzylpiperidine |
| 1-Benzylpiperidin-4-amin |
| N-Benzyl-4-piperidinamine |
| 4-(Piperidin-1-ylmethyl)aniline |
| 1-Benzyl-4-piperidinamine |
| 1-Benzylpiperidin-4-amine |
| 1-(Phenylmethyl)-4-piperidinamine |
| 1-Benzyl-4-aminopiperidine |
| 1-Benzyl -4-amino piperidine |
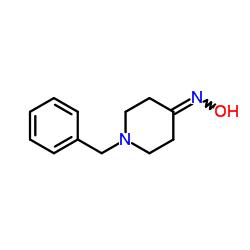 CAS#:949-69-9
CAS#:949-69-9 CAS#:4783-65-7
CAS#:4783-65-7 CAS#:64-17-5
CAS#:64-17-5 CAS#:62992-68-1
CAS#:62992-68-1 CAS#:73889-19-7
CAS#:73889-19-7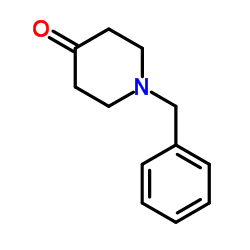 CAS#:3612-20-2
CAS#:3612-20-2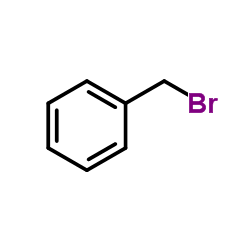 CAS#:100-39-0
CAS#:100-39-0![2-[1-(Phenylmethyl)-4-piperidinyl]-1H-isoindol-1,3(2H)-dione Structure](https://image.chemsrc.com/caspic/132/50534-33-3.png) CAS#:50534-33-3
CAS#:50534-33-3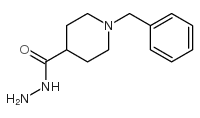 CAS#:74045-91-3
CAS#:74045-91-3 CAS#:100-44-7
CAS#:100-44-7 CAS#:33953-37-6
CAS#:33953-37-6 CAS#:50534-23-1
CAS#:50534-23-1 CAS#:91596-61-1
CAS#:91596-61-1 CAS#:209223-86-9
CAS#:209223-86-9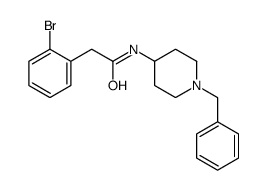 CAS#:208184-69-4
CAS#:208184-69-4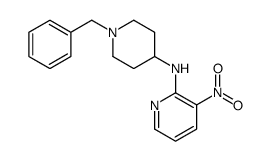 CAS#:185058-54-2
CAS#:185058-54-2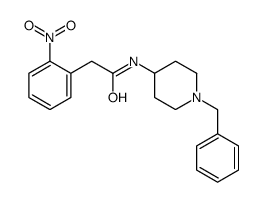 CAS#:208184-80-9
CAS#:208184-80-9![4-(TERT-BUTOXYCARBONYLAMINO)-1-[(6-AMINOPYRIDIN-2-YL)METHYL]PIPERIDINE structure](https://image.chemsrc.com/caspic/005/303763-39-5.png) CAS#:303763-39-5
CAS#:303763-39-5![4-(TERT-BUTOXYCARBONYLAMINO)-1-[(6-BROMOPYRIDIN-2-YL)METHYL]PIPERIDINE structure](https://image.chemsrc.com/caspic/081/303763-37-3.png) CAS#:303763-37-3
CAS#:303763-37-3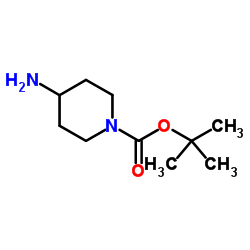 CAS#:87120-72-7
CAS#:87120-72-7
