Cyclooctanone
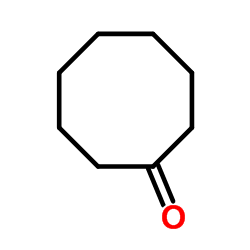
Cyclooctanone structure
|
Common Name | Cyclooctanone | ||
|---|---|---|---|---|
| CAS Number | 502-49-8 | Molecular Weight | 126.196 | |
| Density | 0.9±0.1 g/cm3 | Boiling Point | 193.2±8.0 °C at 760 mmHg | |
| Molecular Formula | C8H14O | Melting Point | 32-41 °C(lit.) | |
| MSDS | Chinese USA | Flash Point | 72.8±0.0 °C | |
| Symbol |

GHS07 |
Signal Word | Warning | |
| Name | Cyclooctanone |
|---|---|
| Synonym | More Synonyms |
| Density | 0.9±0.1 g/cm3 |
|---|---|
| Boiling Point | 193.2±8.0 °C at 760 mmHg |
| Melting Point | 32-41 °C(lit.) |
| Molecular Formula | C8H14O |
| Molecular Weight | 126.196 |
| Flash Point | 72.8±0.0 °C |
| Exact Mass | 126.104462 |
| PSA | 17.07000 |
| LogP | 1.89 |
| Vapour Pressure | 0.5±0.4 mmHg at 25°C |
| Index of Refraction | 1.448 |
| InChIKey | IIRFCWANHMSDCG-UHFFFAOYSA-N |
| SMILES | O=C1CCCCCCC1 |
CHEMICAL IDENTIFICATION
HEALTH HAZARD DATAACUTE TOXICITY DATA
|
| Symbol |

GHS07 |
|---|---|
| Signal Word | Warning |
| Hazard Statements | H315-H319-H335 |
| Precautionary Statements | P261-P305 + P351 + P338 |
| Personal Protective Equipment | Eyeshields;Gloves;type N95 (US);type P1 (EN143) respirator filter |
| Hazard Codes | C:Corrosive; |
| Risk Phrases | R34 |
| Safety Phrases | S45-S36/37/39-S26 |
| RIDADR | 1325.0 |
| WGK Germany | 3 |
| RTECS | GX9800000 |
| Hazard Class | 4.1 |
| HS Code | 2914299000 |
| Precursor 9 | |
|---|---|
| DownStream 10 | |
| HS Code | 2914299000 |
|---|---|
| Summary | 2914299000. other cyclanic, cyclenic or cyclotherpenic ketones without other oxygen function. VAT:17.0%. Tax rebate rate:13.0%. . MFN tariff:5.5%. General tariff:30.0% |
|
Rh(I)-catalyzed intramolecular [3 + 2] cycloaddition of trans-vinylcyclopropane-enes.
J. Am. Chem. Soc. 130(23) , 7178-9, (2008) Vinylcyclopropane (VCP) has been well applied as a five-carbon component, rather than a three-carbon component, in transition-metal catalyzed cycloadditions. Here we demonstrate a Rh(I)-catalyzed [3 +... |
|
|
Total synthesis of (+/-)-asteriscanolide.
J. Org. Chem. 66(22) , 7443-8, (2001) The total synthesis of asteriscanolide (1) has been achieved by taking advantage on an intermolecular Pauson-Khand cycloaddition and a ring-closing metathesis as key bond-forming transformations. The ... |
|
|
Synthesis and antimicrobial evaluation of some new cyclooctanones and cyclooctane-based heterocycles.
Arch. Pharm. (Weinheim) 345(3) , 231-9, (2012) The versatile synthon (E)-2-((dimethyl amino)methylene)cyclooctanone (2) was used as a key intermediate for the synthesis of cyclooctanones and cyclooctane-based heterocycles with pyrazole, isoxazole,... |
| EINECS 207-940-2 |
| MFCD00001754 |
| Cyclooctanone |
| cyclooctan-1-one |
 CAS#:696-71-9
CAS#:696-71-9 CAS#:292-64-8
CAS#:292-64-8 CAS#:931-88-4
CAS#:931-88-4![1,4-Dioxaspiro[4.7]dodecane Structure](https://image.chemsrc.com/caspic/156/183-03-9.png) CAS#:183-03-9
CAS#:183-03-9 CAS#:62249-35-8
CAS#:62249-35-8 CAS#:5452-37-9
CAS#:5452-37-9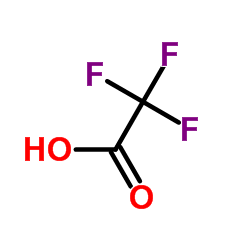 CAS#:76-05-1
CAS#:76-05-1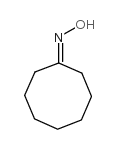 CAS#:1074-51-7
CAS#:1074-51-7![3,3-dimethyl-1,5-dioxaspiro[5.7]tridecane Structure](https://image.chemsrc.com/caspic/254/27889-58-3.png) CAS#:27889-58-3
CAS#:27889-58-3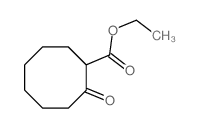 CAS#:4017-56-5
CAS#:4017-56-5 CAS#:5698-29-3
CAS#:5698-29-3 CAS#:496-82-2
CAS#:496-82-2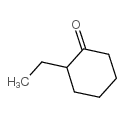 CAS#:4423-94-3
CAS#:4423-94-3 CAS#:3350-30-9
CAS#:3350-30-9 CAS#:1502-06-3
CAS#:1502-06-3 CAS#:3637-63-6
CAS#:3637-63-6![ethyl 2-amino-4,5,6,7,8,9-hexahydrocycloocta[b]thiophene-3-carboxylate structure](https://image.chemsrc.com/caspic/455/40106-16-9.png) CAS#:40106-16-9
CAS#:40106-16-9 CAS#:1459-62-7
CAS#:1459-62-7 CAS#:14519-31-4
CAS#:14519-31-4
