Z-Ile-Ile
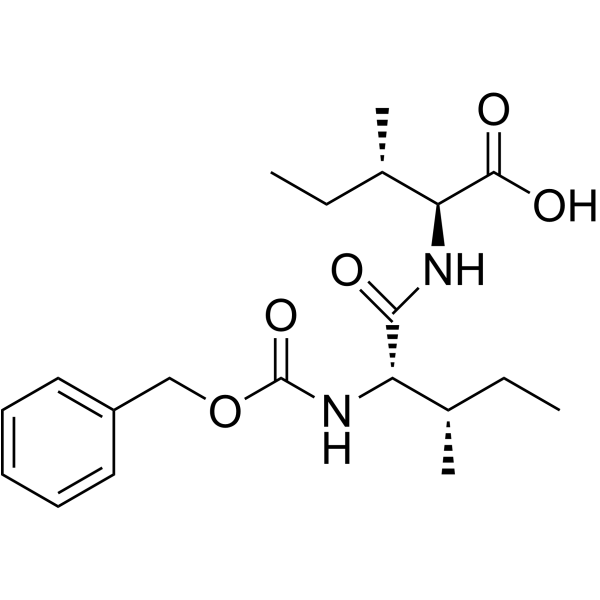
Z-Ile-Ile structure
|
Common Name | Z-Ile-Ile | ||
|---|---|---|---|---|
| CAS Number | 42538-01-2 | Molecular Weight | 378.46300 | |
| Density | 1.13g/cm3 | Boiling Point | 598ºC at 760 mmHg | |
| Molecular Formula | C20H30N2O5 | Melting Point | N/A | |
| MSDS | USA | Flash Point | 315.5ºC | |
Use of Z-Ile-Ile(2S,3S)-2-((2S,3S)-2-(((Benzyloxy)carbonyl)amino)-3-methylpentanamido)-3-methylpentanoic acid is an isoleucine derivative[1]. |
| Name | (2S,3S)-3-methyl-2-[[(2S,3S)-3-methyl-2-(phenylmethoxycarbonylamino)pentanoyl]amino]pentanoic acid |
|---|---|
| Synonym | More Synonyms |
| Description | (2S,3S)-2-((2S,3S)-2-(((Benzyloxy)carbonyl)amino)-3-methylpentanamido)-3-methylpentanoic acid is an isoleucine derivative[1]. |
|---|---|
| Related Catalog | |
| In Vitro | Amino acids and amino acid derivatives have been commercially used as ergogenic supplements. They influence the secretion of anabolic hormones, supply of fuel during exercise, mental performance during stress related tasks and prevent exercise induced muscle damage. They are recognized to be beneficial as ergogenic dietary substances[1]. |
| References |
| Density | 1.13g/cm3 |
|---|---|
| Boiling Point | 598ºC at 760 mmHg |
| Molecular Formula | C20H30N2O5 |
| Molecular Weight | 378.46300 |
| Flash Point | 315.5ºC |
| Exact Mass | 378.21500 |
| PSA | 104.73000 |
| LogP | 3.72480 |
| Index of Refraction | 1.52 |
| InChIKey | QJJULDSUCQXEDB-OTRWWLKZSA-N |
| SMILES | CCC(C)C(NC(=O)C(NC(=O)OCc1ccccc1)C(C)CC)C(=O)O |
| RIDADR | NONH for all modes of transport |
|---|
|
~% 
Z-Ile-Ile CAS#:42538-01-2 |
| Literature: Okumura,Y.; Sakurai,A. Bulletin of the Chemical Society of Japan, 1973 , vol. 46, p. 2190 - 2193 |
|
~% 
Z-Ile-Ile CAS#:42538-01-2 |
| Literature: Okumura,Y.; Sakurai,A. Bulletin of the Chemical Society of Japan, 1973 , vol. 46, p. 2190 - 2193 |
|
~% 
Z-Ile-Ile CAS#:42538-01-2 |
| Literature: Sakurai,A.; Okumura,Y. Bulletin of the Chemical Society of Japan, 1979 , vol. 52, p. 540 - 543 |
| Precursor 3 | |
|---|---|
| DownStream 1 | |
|
Amphipathic property of free thiol group contributes to an increase in the catalytic efficiency of carboxypeptidase Y.
Eur. J. Biochem. 269 , 3220-3225, (2002) Cys341 of carboxypeptidase Y, which constitutes one side of the solvent-accessible surface of the S1 binding pocket, was replaced with Gly, Ser, Asp, Val, Phe or His by site-directed mutagenesis. Kine... |
|
|
Debittering effect of Actinomucor elegans peptidases on soybean protein hydrolysates.
J. Ind. Microbiol. Biotechnol. 35 , 41-47, (2008) Effects of the enzymes in Actinomucor elegans extract and the enzyme Alcalase 2.4L on debittering the soybean protein hydrolysates were investigated. When the protein was treated only with the latter,... |
|
|
Characterization of a bifunctional aminoacylase/carboxypeptidase from radioresistant bacterium Deinococcus radiodurans R1.
J. Biotechnol. 128 , 322-334, (2007) The gene encoding a Deinococcus radiodurans R1 bifunctional aminoacylase/carboxypeptidase (DR_ACY/CP) was amplified by polymerase chain reaction and cloned into pQE-30 to generate pQE-DRAC. The cloned... |
| Z-Ile-Ile-OH |
| N-BENZYLOXYCARBONYL-L-ISOLEUCYL-L-ISOLEUCINE |
| N-CBZ-Ile-Ile |
| Z-Ile-Ile-Oh |
| EINECS 255-876-9 |

 CAS#:42537-99-5
CAS#:42537-99-5
