Pelitinib

Pelitinib structure
|
Common Name | Pelitinib | ||
|---|---|---|---|---|
| CAS Number | 257933-82-7 | Molecular Weight | 467.923 | |
| Density | 1.3±0.1 g/cm3 | Boiling Point | 655.5±55.0 °C at 760 mmHg | |
| Molecular Formula | C24H23ClFN5O2 | Melting Point | 173-178ºC | |
| MSDS | USA | Flash Point | 350.2±31.5 °C | |
| Symbol |

GHS06 |
Signal Word | Danger | |
Use of PelitinibPelitinib (EKB-569;WAY-EKB 569) is an irreversible inhibitor of EGFR with an IC50 of 38.5 nM; also slightly inhibits Src, MEK/ERK and ErbB2 with IC50s of 282, 800, and 1255 nM, respectively. |
| Name | pelitinib |
|---|---|
| Synonym | More Synonyms |
| Description | Pelitinib (EKB-569;WAY-EKB 569) is an irreversible inhibitor of EGFR with an IC50 of 38.5 nM; also slightly inhibits Src, MEK/ERK and ErbB2 with IC50s of 282, 800, and 1255 nM, respectively. |
|---|---|
| Related Catalog | |
| Target |
EGFR:38.5 nM (IC50) |
| In Vitro | Pelitini has much greater inhibitory activity against the EGFR kinase than against Src, MEK/ERK, Cdk4, c-Met, Raf and ErbB2, for example, the IC50 for EGFR is 32-fold lower than the IC50 for the closely related ErbB2. Pelitinib results in a dramatic reduction in EGFR phosphorylation but no change in the total amount of EGFR protein. It requires at least 10-fold more drug to equivalently inhibit ErbB2 phosphorylation in similar assays, and EKB-569 does not block phosphorylation of another receptor tyrosine kinase (c-Met) assessed in the same manner[1]. EKB-569 is a potent inhibitor of proliferation in NHEK, A431, and MDA-468 cells (IC50=61, 125, and 260 nM, respectively) but not MCF-7 cells (IC50=3600 nM). EKB-569 is also a potent inhibitor of EGF-induced phosphorylated EGF-R (pEGF-R) in A431 and NHEK cells (IC50=20-80 nM)[1]. |
| In Vivo | A single oral dose of 10 mg/kg EKB-569 inhibits EGFR phosphorylation in A431 xenografts within 60 minutes. Twenty-four hours later, EGFR activity is still inhibited by over 50% by this single oral dose. The half-life of EKB-569 in mouse plasma is about 2 hours[1]. |
| Cell Assay | For experiments using cells in culture, A431 cells or 3T3/c-erbB-2 cells over-expressing c-erbB2 are are treated with various concentrations of EKB-569 for 2.75 h before co-incubation with 100 ng/mL EGF (A431 cells) or no growth factor (3T3/c-erbB-2 cells) for 0.25 h. Cells are ished twice with cold phosphate-buffered saline (PBS) before adding to lysis buffer for 20 min on ice, before immunoprecipitation and SDS–PAGE-immunoblotting[1]. |
| Animal Admin | Mice: For in vivo experiments, athymic nu/nu female mice are implanted subcutaneously with 5×106 A431 tumor cells. When tumors reach a mass of 200-300 mg, animals are treated with a single dose of 10 mg/kg EKB-569 in pH 2.0 water per gavage. Tumors from control and drug-treated animals are excised and minced into 1-mm pieces for anlysis[1]. |
| References |
| Density | 1.3±0.1 g/cm3 |
|---|---|
| Boiling Point | 655.5±55.0 °C at 760 mmHg |
| Melting Point | 173-178ºC |
| Molecular Formula | C24H23ClFN5O2 |
| Molecular Weight | 467.923 |
| Flash Point | 350.2±31.5 °C |
| Exact Mass | 467.152435 |
| PSA | 90.28000 |
| LogP | 5.95 |
| Vapour Pressure | 0.0±2.0 mmHg at 25°C |
| Index of Refraction | 1.645 |
| Storage condition | -20°C Freezer |
| Water Solubility | DMSO: soluble5mg/mL, clear (warmed) |
| Precursor 9 | |
|---|---|
| DownStream 0 | |
|
Sensitivity of Melanoma Cells to EGFR and FGFR Activation but Not Inhibition is Influenced by Oncogenic BRAF and NRAS Mutations.
Pathol. Oncol. Res. 21 , 957-68, (2015) BRAF and NRAS are the two most frequent oncogenic driver mutations in melanoma and are pivotal components of both the EGF and FGF signaling network. Accordingly, we investigated the effect of BRAF and... |
|
|
Pelitinib (EKB-569) targets the up-regulation of ABCB1 and ABCG2 induced by hyperthermia to eradicate lung cancer.
Br. J. Pharmacol. 172 , 4089-106, (2015) Pelitinib is a potent irreversible EGFR TK inhibitor currently in clinical trials for the treatment of lung cancer. Hyperthermia has been applied concomitantly with chemotherapy and radiotherapy to en... |
|
|
A molecular case report: functional assay of tyrosine kinase inhibitors in cells from a patient's primary renal cell carcinoma.
Cancer Biol. Ther. 14 , 95-9, (2013) Current therapies for Renal Cell Carcinoma favor vascular endothelial growth factor receptor (VEGF-R) tyrosine kinase (TK) inhibitors (TKIs). In theory, these are most applicable in tumors that have l... |
| 2-Butenamide,N-[4-[(3-chloro-4-fluorophenyl)amino]-3-cyano-7-ethoxy-6-quinolinyl]-4-(dimethylamino)-,(2E) |
| 4-dimethylamino-but-2-enoic acid [4-(3-chloro-4-fluoro-phenylamino)-3-cyano-7-ethoxy-quinolin-6-yl]-amide |
| 3-Cyano-4-[(3-chlor-4-fluorphenyl)amino]-6-{[4-(N,N-dimethylamino)-1-oxo-2-buten-1-yl]amino}-7-ethoxy-chinoline |
| (2E)-N-[4-[(3-Chloro-4-fluorophenyl)amino]-3-cyano-7-ethoxy-6-quinolinyl]-4-(dimethylamino)-2-butenamide |
| (2E)-N-{4-[(3-Chloro-4-fluorophenyl)amino]-3-cyano-7-ethoxyquinolin-6-yl}-4-(dimethylamino)but-2-enamide |
| pelitinib |
| (2E)-N-[4-[(3-chloro-4-fluorophenyl)amino]-3-cyano-7-ethoxy-6-quinolinyl]-4-(dimethylamino)-2-buteneamide |
| WAY-EKB 569 |
| EKB-569 |
| (E)-N-(4-(3-chloro-4-fluorophenylamino)-3-cyano-7-ethoxyquinolin-6-yl)-4-(dimethylamino)but-2-enamide |
| (2E)-N-{4-[(3-Chloro-4-fluorophenyl)amino]-3-cyano-7-ethoxy-6-quinolinyl}-4-(dimethylamino)-2-butenamide |
| 2-Butenamide, N-[4-[(3-chloro-4-fluorophenyl)amino]-3-cyano-7-ethoxy-6-quinolinyl]-4-(dimethylamino)-, (2E)- |
| 3-cyano-4-[(3-chloro-4-fluorophenyl)amino]-6-{[4-(N,N-dimethylamino)-1-oxo-2-buten-1-yl]amino}-7-ethoxy-quinoline |
 CAS#:124-40-3
CAS#:124-40-3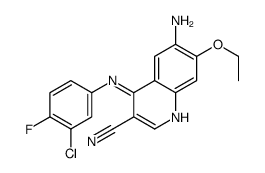 CAS#:361162-95-0
CAS#:361162-95-0 CAS#:367-21-5
CAS#:367-21-5 CAS#:106579-00-4
CAS#:106579-00-4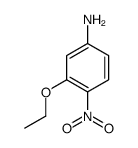 CAS#:116435-75-7
CAS#:116435-75-7 CAS#:361162-90-5
CAS#:361162-90-5 CAS#:214476-07-0
CAS#:214476-07-0 CAS#:361162-91-6
CAS#:361162-91-6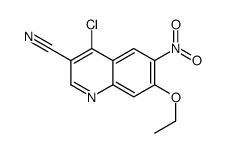 CAS#:214476-09-2
CAS#:214476-09-2
